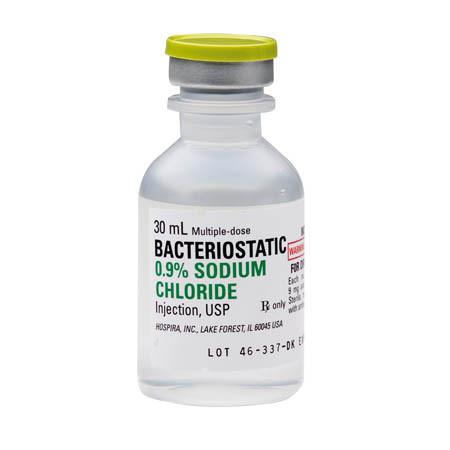
Bacteriostatic Sodium Chloride 0.9% Injection 30ml Multiple Dose Vials, 25/Pack (Rx)
(Note: We don’t Fill Personal Prescriptions)
How to Order:
Bacteriostatic Sodium Chloride 0.9% Injection 30ml Multiple Dose Vials is an effective agent for inhibiting the growth of bacteria for medical use. It is a sterile injection which is used to help support the function of kidneys or as an aid in respiratory organ ailments. It helps maintain the balance of the pH and osmolality in intravenous solutions. Bacteriostatic Sodium Chloride can help prevent an infection or stop an existing infection after being introduced into the body. Its use varies from rehydration to diluting vaccinations, and it can be virtually used for experimenting in various clinical settings. Most importantly, Bacteriostatic Sodium Chloride 0.9% Injection 30ml Multiple Dose Vials can help assist in medical procedures more effectively than a regular saline solution because of its Bacteriostatic Sodium Chloride component.
Bacteriostatic 0.9% Sodium Chloride Injection 30ml is a sterile, nonpyrogenic, hypotonic solution of sodium chloride in water for injection. It is intended for intravenous use. It contains no bacteriostatic or antimicrobial agent and is unfit for parenteral injection. Sodium chloride 0.9% is a diluent and osmotic agent which exerts its principal effect by causing water to flow into the cells of the body. The intracellular fluid compartment is increased, resulting in a decrease in serum osmolality and an increase in the extracellular fluid volume. This action occurs because the sodium chloride molecule is smaller than the water molecule and is thus able to pass through the cell membrane. Bacteriostatic 0.9% Sodium Chloride Injection 30ml is indicated for the temporary restoration of volumetric deficit in adults.
Benefits of Bacteriostatic Sodium Chloride 0.9% Injection
- Highly effective in controlling the growth and spread of bacteria
- Low risk of side effects, since it is a simple saline solution
- Preservative-free for maximum safety
- Aids in wound healing and skin hydration
- Easy to store and transport
- Versatile application for multiple uses including irrigation, flushing, and eye irrigation
- Reliable potency and quality assurance
- Reduced risk of contamination risks due to the vials disposable design
- Cost-effective option for bulk purchasing
- Non-pyrogenic, eliminating potential for hazardous reactions associated with other medications
Instructions on How to Use Bacteriostatic Sodium Chloride 0.9% Injection
1. Wash your hands thoroughly and put on sterile gloves.
2. Look at the solution in the container and check for wispy particles. Do not use the solution if you find any.
3. loosen the protective cap on the container and insert the administration set septum protector into the septum.
4. Attach the infusion line and activate the flow.
5. Verify the volume of the correct solution and begin infusion.
6. Hang the solution container next to the patients bed at a height that allows an unobstructed line of sight to determine how much fluid is being infused.
7. Monitor the infusion rate for individual patient needs.
8. When infusion is complete, put the protective cap on the container and remove the line from the set.
9. Discard the solution and materials as directed by your require.
10. Wash your hands with soap and water again.
Pfizer Injectables Bacteriostatic 0.9% Sodium Chloride Injection 30ml is a sterile, multiple-dose nonpyrogenic, isotonic solution of sodium chloride (normal saline) in water for injection.
Each milliliter (mL) contains sodium chloride 9 mg and 0.9% (9 mg/mL) benzyl alcohol as a bacteriostatic preservative. May contain hydrochloric acid to balance/ adjust pH level. It is supplied in a multiple-dose flip-top vial that can be used for repeated withdrawals to dilute or dissolve drugs for medication. The pH is 5.0 (4.5 to 7.0).
NDC: 0409-1966-07
UPC: 00304091966024
Strength: 0.9%
Count: 25 Multiple Dose Vials