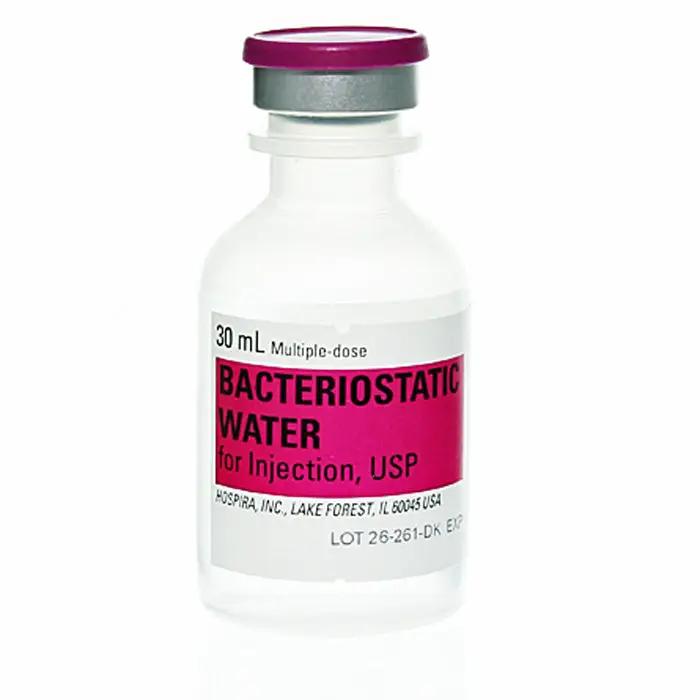
Bacteriostatic Water for Injection (Sterile Water) Multiple Dose Vials 30mL, 25/Pack (Rx)
(Note: We don’t Fill Personal Prescriptions)
How to Order:
Bacteriostatic water is a sterile water solution that contains a small amount of benzyl alcohol, which prevents the growth of bacteria. Bacteriostatic water is commonly used in medical and research settings to store and dilute medications or reagents for injection or topical application. Bacteriostatic water is also used to reconstitute certain medications in powder form before injection, as well as for diluting and storing human chorionic gonadotropin (HCG) for hormone therapy. Its sterile and bacteriostatic properties make it an important component in the safe administration of various medications.
Pfizer Injectables Bacteriostatic Water for Injection is sterile, non-pyrogenic water that contains 0.9% benzyl alcohol (9mg/mL) added as a bacteriostatic preservative. This preparation of sterile water and benzyl alcohol (BnOH) allows repeated withdrawals to be made from a single 30mL plastic vial. Hospira, the world’s leading manufacturer and supplier of bacteriostatic and sterile water, advises that once a vial of bacteriostatic water is open, the preservatives will prolong its life for up to 28 days. After this period, the vial should be discarded.
Bacteriostatic Water for Injection is a crucial and versatile solution used in medical settings to prepare various pharmaceutical medications. This sterile water contains a small amount of benzyl alcohol, which helps to inhibit the growth of bacteria and fungi, making it safe for multiple uses. Bacteriostatic Water is commonly used to dilute and dissolve medications that are given via injections, such as antibiotics, hormones, and vaccines. Its key role in safeguarding against the growth of harmful microorganisms ensures medication potency and safety. Bacteriostatic Water's unique features, including its sterile properties and ability to prevent bacterial growth, make it a preferred choice in clinics, hospitals, and even in-home healthcare settings. This essential solution offers multiple benefits, from ensuring medication stability to reducing the risk of infection during injections. Overall, the use of Bacteriostatic Water empowers healthcare providers to deliver top-quality care and ensures safety for patients, making it a vital product in the medical field..
The difference between bacteriostatic water and sterile water is that other sterile waters for injection do not contain a bacteriostatic agent. As such, they are single-dose vials and should be discarded after a single use.
Bacteriostatic Water for Injection, USP is a sterile, non-pyrogenic, sterile water that contains 0.9% benzyl alcohol (9mg/mL) added as a bacteriostatic preservative. Bacteriostatic water can be used multiple times up to 28 days once opened. Pfizer recommends that after this time you must discard the vial.This preparation of sterile water and benzyl alcohol (BnOH) allows repeated withdrawals to be made from a single 30mL vial. Hospira Worldwide now Pfizer injectables is the world’s leading manufacturer of bacteriostatic water, Pfizer injectables recommends that once the vial of bacteriostatic water is open the preservatives will prolong its life for up to 28 days and that after this period of time the vial should to be discarded.
A bacteriostatic agent or bacteriostat is a biological or chemical agent that stops bacteria from reproducing, while not necessarily killing them otherwise. Depending on their application, bacteriostatic antibiotics, disinfectants, antiseptics and preservatives can be distinguished. When bacteriostatic antimicrobials are used, the duration of therapy must be sufficient to allow host defense mechanisms to eradicate the bacteria. Upon removal of the bacteriostat, the bacteria usually start to grow again. This is in contrast to bactericides, which kill bacteria.
How to Mix medications with Bacteriostatic water:
- Using an alcohol pad, wipe the rubber stopper on the first vial to decrease the risk of contaminating the medication as you insert the needle into the vial.
- Pull back syringe plunger until the volume of air drawn into the syringe equals the volume to be withdrawn from the drug vial.
- Without inverting the vial, insert the needle into the top of the vial, making sure that the needle's bevel tip doesn't touch the solution. Inject the air into the vial and withdraw the needle. This step replaces air in the vial, thus preventing the creation of a partial vacuum on the withdrawal of the drug.
- With a new needle, repeat the step above for the second vial or empty mixing vial. Then, after injecting the air into the second vial, invert the vial, withdraw the prescribed dose, and then withdraw the needle (as shown below).
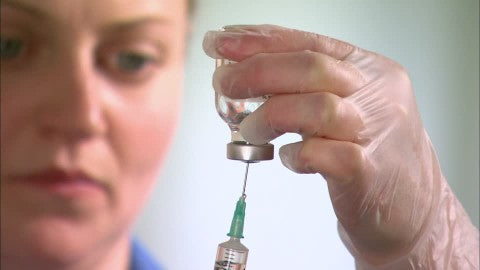
- Wipe the rubber stopper of the first vial again and insert the needle, taking care not to depress the plunger. Invert the vial, withdraw the prescribed dose, and then withdraw the needle.
- Change the needle on the syringe, if indicated.
- Last, discard all waste in appropriate sharps container of waste receptacle.
Complications
Complications, such as adverse drug reaction, may occur if an incorrect dose of medication is prepared or incompatible medications are mixed and administered.
What is Bacteriostatic Water?
Bacteriostatic Water for Injection is sterile non-pyrogenic water that contains 0.9% benzyl alcohol (9mg/mL), which is added as a bacteriostatic preservative. This preparation of sterile water and benzyl alcohol (BnOH) allows repeated withdrawals to be made from a single 30mL plastic vial. Hospira, the world’s leading manufacturer and supplier of bacteriostatic and sterile water, recommends that once a vial of bacteriostatic water is open the preservatives will prolong its life for up to 28 days and that after this period of time the vial should to be discarded. Other sterile waters for injection without the bacteriostatic agent (single-dose vials) should be discarded after a single use. Visit our page to purchase online at wholesale supply prices.
The semi-rigid vial is fabricated from a specially formulated polyolefin. It is a copolymer of ethylene and propylene. The safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers. The container requires no vapor barrier to maintain the proper labeled volume.
Bacteriostatic Water MSDS Sheets
Do not use Bacteriostatic Water for Injection, USP for intravenous injection unless the osmolar concentration of additives results in an approximate isotonic admixture.
Consult the manufacturer’s instructions for choice of vehicle, appropriate dilution or volume for dissolving the drugs to be injected, including the route and rate of injection.
Inspect reconstituted (diluted or dissolved) drugs for clarity (if soluble) and freedom from unexpected precipitation or discoloration prior to administration.
Pregnancy Category C. Animal reproduction studies have not been conducted with Bacteriostatic Water for Injection. It is also not known whether Bacteriostatic Water for Injection containing additives can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Bacteriostatic Water for Injection, USP containing additives should be given to a pregnant woman only if clearly needed.
Pediatric Use:
The safety and effectiveness of Bacteriostatic Water for Injection, USP have not been established in pediatric patients. Due to the potential for toxicity, solutions containing benzyl alcohol should not be used in neonates.
Drug Interactions:
Some drugs for injection may be incompatible in a given vehicle, or when combined in the same vehicle or in a vehicle containing benzyl alcohol. Consult with pharmacist, if available.
Use aseptic technique for single or multiple entry and withdrawal from all containers.
When diluting or dissolving drugs, mix thoroughly and use promptly.
Do not store reconstituted solutions of drugs for injection unless otherwise directed by the manufacturer of the solute.
Do not use unless the solution is clear and seal intact.
ADVERSE REACTIONSReactions which may occur because of this solution, added drugs or the technique of reconstitution or administration include febrile response, local tenderness, abscess, tissue necrosis or infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection and extravasation.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate countermeasures, and if possible, retrieve and save the remainder of the unused vehicle for examination.
Although adverse reactions to intravenous, intramuscular or subcutaneous injection of 0.9% benzyl alcohol are not known to occur in man, experimental studies of small volume parenteral preparations containing 0.9% benzyl alcohol in several species of animals have indicated that an estimated intravenous dose up to 30 mL may be safely given to an adult without toxic effects. Administration of an estimated 9 mL to a 6 kg infant or neonate is potentially capable of producing blood pressure changes.
DOSAGE AND ADMINISTRATIONThe volume of the preparation to be used for diluting or dissolving any drug for injection is dependent on the vehicle concentration, dose and route of administration as recommended by the manufacturer.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit Instructions for Use: First examine vial appearance. Make sure sterile bacteriostatic water inside is clear and seal is intact and undamaged. Purple plastic flip top helps protect rubber injection entry site.
Swab top of rubber injection site with antiseptic wipe containing 70% isopropyl alcohol or higher to reduce any existing bacteria growth. Wait until alcohol is dry before continuing.
Next, using a sterile syringe and wearing sterile examination gloves, draw the exact dose of sterile water from the vial and carefully injected it into an empty vial to prepare for mixing. Mix thoroughly with desired drug and use promptly after mixing.
After the first time the water is used, label the vial with date and time to ensure preservative integrity (25-28 days once opened). Store at 20 to 25 Celsius (68 to 77 degrees Fahrenheit).
How long does it last once it's opened? Hospira recommends that once a vial open you have up to 28 days to use it. After 28 days the vial needs to be discarded. Sterile water for injection without benzyl alcohol added (single-dose vials) should be discarded after one single use. Sterile Water for injection is single-dose sterile water that does not contain any Bacteriostatic, antimicrobial agents, and added buffers.Store: 20° to 25°C (68 to 77°F) Not for use in neonates. See package label for additional warnings and precautions prior to use. Not recommended on neonates because on the toxicity of the preservative benzyl alcohol. Bacteriostatic water.