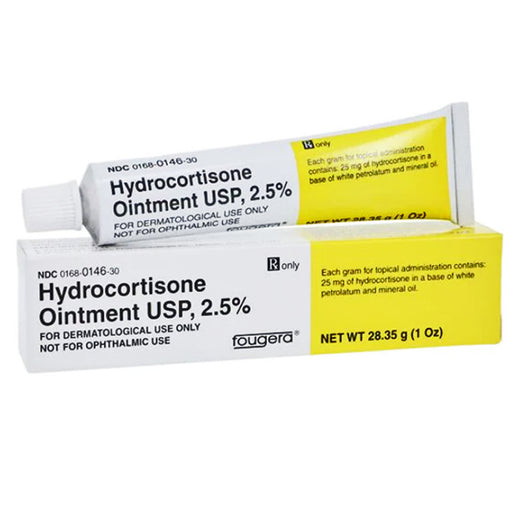
Triamcinolone Acetonide Ointment 0.1% (Aristocort) by Perrigo is used to reduce the actions of chemicals in the body that cause inflammation, redness, and swelling. Triamcinolone topical is used to treat the inflammation caused by several conditions, such as allergic reactions, eczema, and psoriasis. The dental paste form of triamcinolone is used to treat mouth ulcers.
Use this medication only on the skin. Do not use it on the face, groin, or underarms unless directed to do so by your doctor.
Wash and dry your hands. Before applying the medication, clean and dry the affected area. Apply a thin film of the medication to the affected area and gently rub in, usually 2 to 4 times daily or as directed by your doctor. Do not cover, bandage or wrap the area unless directed to do so by your doctor. If used in the diaper area on an infant, do not use tight-fitting diapers or plastic pants.
After applying the medication, wash your hands unless you are using this medication to treat the hands. When applying this medication near the eyes, avoid getting it in the eyes as this may worsen or cause glaucoma. Also, avoid getting this medication in the nose or mouth. If you get the medication in your eyes, nose, or mouth, rinse with plenty of water.
Brand: Perrigo
Size: 454 grams (1 Pound)
NDC: 45802-0055-05
UPC: 345802055057
Buy Perrigo Triamcinolone Acetonide Ointment 0.1% Jar, 1 Pound online at Mountainside Medical Equipment.
- Product: Triamcinolone Acetonide Ointment USP 0.1% in a 454g (1 pound) jar — a medium-potency topical corticosteroid in a petrolatum-based, occlusive ointment vehicle, manufactured by Perrigo / Padagis (NDC: 45802-0055-05 | UPC: 345802055057)
- Manufacturer note: Perrigo rebranded its generic Rx pharmaceutical division as Padagis; both names refer to the same manufacturer and product — NDC 45802-0055-05 is the definitive identifier for procurement and formulary records
- Large-format advantage: The 454g triamcinolone acetonide ointment jar is one of the largest commercially available single-unit sizes, designed for high-volume in-office dispensing, institutional procurement, and bulk clinical supply — distinct from standard 15g, 30g, or 80g tube sizes intended for individual patient use
- Clinical positioning: Classified as a Group IV–V (medium-potency) topical corticosteroid; designed to support management of corticosteroid-responsive inflammatory dermatoses, including eczema, psoriasis, and allergic skin reactions — for use by licensed healthcare professionals only
- Formulation distinction: The petrolatum-based ointment vehicle provides enhanced skin penetration compared to cream formulations, making it the preferred clinical format for dry, thickened, lichenified, or scaly skin presentations
- Wholesale access: Available at competitive wholesale pricing through Mountainside Medical Equipment; free US shipping on orders over $100; new accounts receive 5% off their first purchase
- Licence requirement: This is a prescription-only product; purchase requires a valid medical, pharmaceutical, or institutional licence — not available over the counter
| Specification | Detail |
|---|---|
| Generic name | Triamcinolone Acetonide Ointment USP 0.1% |
| Brand reference | Aristocort (originator brand; this is a Perrigo / Padagis generic equivalent) |
| Manufacturer | Perrigo / Padagis |
| NDC number | 45802-0055-05 |
| UPC | 345802055057 |
| Concentration | 0.1% (1mg triamcinolone acetonide per gram of ointment) |
| Net weight / jar size | 454g (1 pound) |
| Formulation type | Ointment — petrolatum-based, occlusive vehicle |
| USP designation | USP monograph compliant |
| Potency classification | Medium-potency topical corticosteroid (Group IV–V, US classification) |
| Drug class | Topical corticosteroid/anti-inflammatory |
| Route of administration | Topical (external skin surface only) |
| Typical application frequency | 2 to 4 times daily or as directed by the prescribing clinician |
| Storage conditions | Not specified in the provided data |
| Country of supply | United States |
- FDA-approved indication: Intended to support relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses, per FDA-approved prescribing information for triamcinolone acetonide topical formulations (ref: DailyMed NDC 45802-0055-05)
- Clinical applications: Commonly applied in dermatology, primary care, wound care, and medspa settings to support management of atopic dermatitis (eczema), plaque psoriasis, contact dermatitis, seborrheic dermatitis, and allergic inflammatory skin conditions in patients under licensed clinical care
- Formulation selection: The ointment vehicle is the clinically preferred format for dry, thickened, lichenified, or scaly lesions; the occlusive petrolatum base enhances corticosteroid penetration into the stratum corneum — cream formulations are more appropriate for moist, acutely inflamed, or intertriginous presentations
- Application protocol: Apply a thin film to the clean, dry affected skin area 2 to 4 times daily or as directed by the prescribing clinician; do not bandage, wrap, or occlude the treated area unless specifically directed by the clinician
- Anatomical restrictions: Not intended for application to the face, groin, or underarm areas unless specifically directed by a licensed prescriber; these regions have thinner skin and higher percutaneous absorption rates, increasing risk of local and systemic adverse effects
- Paediatric use: When applied in the diaper area of infants, tight-fitting diapers or occlusive plastic pants must not be used — occlusion significantly increases systemic absorption of the corticosteroid in this population
- Professional oversight required: For use by or under the direction of a licensed healthcare professional; clinicians must consult the full FDA-approved prescribing information and package insert before clinical application
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
- Prescription-only product: Triamcinolone acetonide ointment USP 0.1% is not approved for over-the-counter use in the United States; it requires a valid prescription and must be used under the direction of a licensed prescriber
- Restricted anatomical sites: Do not apply to the face, groin, or underarm areas unless specifically directed by a licensed clinician; application to thin or sensitive skin carries a significantly elevated risk of skin atrophy, telangiectasia, and striae
- Ocular exposure risk: Avoid contact with the eyes; inadvertent ophthalmic exposure may worsen pre-existing glaucoma or precipitate new-onset glaucoma or cataracts with prolonged near-eye application; if eye contact occurs, rinse immediately and thoroughly with water
- Systemic absorption and HPA axis suppression: Prolonged use, large surface area application, or use under occlusive dressings may result in HPA axis suppression, Cushing's syndrome features, or hyperglycaemia — risks are elevated in paediatric patients, patients with compromised skin barriers, and those treated with occlusion
- Paediatric occlusion warning: In infants and young children, do not use tight-fitting diapers or occlusive plastic pants over treated skin areas; occlusion dramatically increases percutaneous absorption and systemic exposure to the corticosteroid
- Infection contraindication: Do not apply to untreated bacterial, fungal, or viral skin infections; topical corticosteroids may mask signs of infection and suppress local immune response, potentially worsening an underlying infectious process
- Full prescribing information: This safety summary does not replace the complete FDA-approved prescribing information; all prescribing clinicians and dispensing professionals must consult the full label, available via DailyMed (NDC: 45802-0055-05), before clinical use
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
- Licence requirement: Purchase of triamcinolone acetonide ointment USP 0.1% from Mountainside Medical Equipment requires a valid medical, pharmaceutical, or institutional licence appropriate to the buyer's jurisdiction; this product is not available to the general public or for over-the-counter resale
- Eligible buyer types: Licensed clinics, hospitals, dermatology and primary care practices, medspas and aesthetic professionals, dental offices, EMS and emergency response teams, veterinary practices, and qualified institutional procurement departments across the United States
- Wholesale pricing: Mountainside Medical offers competitive wholesale pricing on the triamcinolone acetonide ointment jar and all stocked pharmaceutical products; volume and bulk order pricing is available upon enquiry
- New account discount: New customers receive 5% off their first purchase at Mountainside Medical Equipment
- Shipping: Free US shipping on all orders over $100; Mountainside Medical ships across the United States — contact the sales team for specific delivery timeline information
- Contact for orders and enquiries: Phone: +1 (888) 687-4334 | Email: sales@mountainside-medical.com — the sales team assists with bulk order quotes, licence verification, account setup, and stock availability confirmation
- Stock confirmation: Contact Mountainside Medical directly to verify current stock availability for the 454g triamcinolone acetonide ointment jar (NDC: 45802-0055-05) before placing a bulk or volume order
Professional Use Only: Triamcinolone Acetonide Ointment USP 0.1% is intended for purchase and use by licensed healthcare professionals only. This product requires a valid prescription and is not approved for over-the-counter sale or use without the supervision of a licensed prescriber.
Clinical Information Disclaimer: The clinical and pharmaceutical information provided on this page is intended for reference purposes by qualified healthcare professionals only. It does not constitute medical advice, a clinical recommendation, or a substitute for the full FDA-approved prescribing information. All prescribing clinicians and dispensing professionals must consult the complete package insert and FDA-approved label before clinical use. The full prescribing information for this product is accessible via the US National Library of Medicine DailyMed database (NDC: 45802-0055-05).
Regulatory Reference: All indications referenced on this page reflect FDA-approved prescribing information for triamcinolone acetonide topical formulations as documented by the National Library of Medicine DailyMed database. Mountainside Medical Equipment does not make independent clinical efficacy or safety claims beyond those supported by the FDA-approved label for this product.
Manufacturer Information: This product is manufactured by Perrigo / Padagis. Mountainside Medical Equipment is a wholesale distributor and does not manufacture, reformulate, or repackage this product. Manufacturer specifications, quality standards, and regulatory standing are as represented by the manufacturer and identified by NDC 45802-0055-05. No additional manufacturer quality, country of origin, or regulatory claims beyond those present in the provided product data are made or implied.
For clinical guidance, consult a licensed pharmacist or physician. For ordering, licensing, and account enquiries, contact Mountainside Medical Equipment at +1 (888) 687-4334 or sales@mountainside-medical.com.
-
What is triamcinolone acetonide ointment USP 0.1% used for?
Triamcinolone acetonide ointment USP 0.1% is a medium-potency topical corticosteroid indicated — per FDA-approved prescribing information — for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. In clinical practice, licensed healthcare professionals use it to support management of atopic dermatitis (eczema), plaque psoriasis, contact dermatitis, seborrheic dermatitis, and related allergic inflammatory skin conditions. It is a prescription-only product and is not for over-the-counter use.
-
What is the difference between triamcinolone acetonide ointment and triamcinolone acetonide cream?
The ointment and cream formulations of triamcinolone acetonide 0.1% differ in their vehicle base, skin penetration depth, and clinical suitability. The ointment uses a petrolatum-based occlusive vehicle that enhances corticosteroid absorption into the stratum corneum, making it the preferred clinical choice for dry, thickened, lichenified, or scaly lesions such as chronic plaque psoriasis or lichenified eczema. The cream uses a water-in-oil emulsion vehicle that is less occlusive and more cosmetically acceptable, making it better suited to moist, acutely inflamed, or intertriginous areas. Prescribing clinicians should select the formulation based on the clinical presentation and lesion morphology.
-
Why does this product show both Perrigo and Padagis branding?
Perrigo completed a strategic divestiture of its generic Rx pharmaceutical manufacturing division, which was subsequently rebranded as Padagis. Both names refer to the same manufacturer and the same product line. NDC 45802-0055-05 is the authoritative identifier for this specific product — Triamcinolone Acetonide Ointment USP 0.1% in the 454g jar — and remains consistent across the rebrand. Procurement officers and formulary managers can use this NDC to confirm product identity in any dispensing or procurement system.
-
What is the NDC for the 1 pound triamcinolone acetonide ointment jar?
The NDC (National Drug Code) for Triamcinolone Acetonide Ointment USP 0.1% in the 454g (1 pound) jar manufactured by Perrigo / Padagis is 45802-0055-05. The UPC is 345802055057. These identifiers should be used for all procurement records, formulary submissions, pharmacy system entry, and dispensing documentation.
-
Is triamcinolone acetonide ointment 0.1% available over the counter?
No. Triamcinolone acetonide ointment USP 0.1% is a prescription-only pharmaceutical product in the United States and is not approved for over-the-counter sale. Mountainside Medical Equipment supplies this product exclusively to licensed healthcare professionals and qualified institutional buyers. A valid medical, pharmaceutical, or institutional licence is required to complete a purchase. This requirement ensures clinical oversight of a corticosteroid that carries documented risks — including HPA axis suppression and skin atrophy — when used outside professional supervision.
-
Why is the 454g triamcinolone acetonide ointment jar used in clinical and institutional settings?
The 1 pound (454g) triamcinolone acetonide ointment jar is designed for high-volume clinical use, in-office dispensing, and institutional procurement. Standard tube sizes — typically 15g, 30g, or 80g — are intended for individual patient dispensing. The large-format jar reduces per-unit cost, minimises reorder frequency, and supports efficient dispensing workflows in facilities treating high patient volumes. Dermatology practices, wound care centres, medspas managing post-procedure inflammation, and institutional buyers commonly procure this format for these operational and cost reasons.
-
Can triamcinolone acetonide ointment be applied to the face?
Triamcinolone acetonide ointment USP 0.1% should not be applied to the face, groin, or underarm areas unless a licensed prescriber has specifically directed its use in those locations. These anatomical regions feature thinner skin with higher percutaneous absorption rates, which significantly increases the risk of local adverse effects including skin atrophy, telangiectasia, and striae. Prescribing clinicians should consult the full FDA-approved prescribing information before directing use at any sensitive or restricted anatomical site.
-
How do I order the triamcinolone acetonide ointment jar at wholesale pricing?
Mountainside Medical Equipment offers wholesale pricing on Triamcinolone Acetonide Ointment USP 0.1% (454g jar, NDC: 45802-0055-05) to licensed clinics, hospitals, dermatology practices, medspas, EMS teams, dental offices, and institutional buyers across the United States. To place an order, request a volume pricing quote, or complete licence verification, contact the Mountainside Medical sales team at +1 (888) 687-4334 or sales@mountainside-medical.com. Free shipping applies to US orders over $100, and new accounts receive 5% off their first purchase.