Filters
- 29085 (1)
- 60 Drop (1)
- 60 drop iv set (1)
- 60 Drop Microdrip (1)
- Exel iv sets (1)
- IV Administration Set (1)
- IV Set (1)
- IV Set 60 drop (1)
- Iv Sets (1)
- IV Tubing (1)
- Luer Lock (1)
- 11948-02 (1)
- 60 drip (1)
- Burettes Clave (1)
- clave injection site (1)
- lifeshield (2)
- microdrip (2)
- plum set (1)
- soluset (1)
- 14270-28 (1)
- macroboro (1)
IV Sets, IV Tubing, IV Administration Sets to microdrip

IV Sets
IV sets (administration sets) are sterile, flexible plastic tubing systems used to deliver fluids, medications, or blood products directly into a patient’s vein. Key components include a spike for the bag, a drip chamber, a flow regulator (clamp), and a Luer connector. They are crucial for maintaining precise fluid administration and preventing air from entering the blood stream.
An IV set (or administration set) is a sterile, single-use plastic assembly that connects a fluid source (IV bag or bottle, blood unit, etc.) to a patient’s intravenous catheter or needle. The basic components include a rigid spike (to puncture the IV bag port), an air-vent or filter, a clear drip chamber, flexible tubing with clamps or flow regulators, one or more injection ports (often needleless) for meds, and a Luer-lock (or slip) connector at the patient end. For example, parts are typically named “spike, air vent cap, drip chamber (macro or micro), fluid filter, flow regulator (roller clamp or pinch clamp), injection site (Y-site with needle-free connector), and Luer connector”. Some sets also include built-in filters (e.g. 0.2 μm) or burette chambers for precise dosing. IV sets are always packaged sterile and used once – after priming and use they are discarded as medical sharps to prevent contamination. Proper aseptic technique is essential: all connection points (spike, ports, catheter) must remain sterile to avoid introducing pathogens. (Indeed, infection-control guidelines emphasize that IV delivery systems are sterile single-use devices and strict asepsis must be maintained during setup and use.
Sizes and Flow (Drop Factors)
IV tubing comes in various “drop factors” that define how many drops make 1 mL of fluid. Common designs are:
- Macrodrip (10–20 gtt/mL) – This is a larger-bore tubing where 1 mL ≈ 10–20 drops. Macrodrip sets are used for adult infusions or rapid fluid replacement (e.g. large-volume resuscitation). For example, a 15 gtt/mL set means ~15 drops = 1 mL.
- Microdrip (60 gtt/mL) – This fine-bore tubing yields 60 drops = 1 mL (each drop ≈0.017 mL), allowing very precise control. Microdrip sets are typically used for low-rate infusions (pediatrics, neonates, critical care) where accuracy matters.
Because microdrip sets produce tiny droplets, they are best when infusion rates are slow (commonly < 50–100 mL/hr) so that counting drops is feasible; macrodrip sets are preferred for higher rates (e.g. 100–2000 mL/hr) since fewer drops per mL can be counted or delivered more rapidly. To calculate flow:
- Drops per minute = (Volume (mL) to infuse × Drop factor (gtt/mL)) ÷ Time (min).
For instance, infusing 100 mL over 1 hour with a 15 gtt/mL set: (100 mL × 15 gtt/mL) ÷ 60 min ≈ 25 drops/min. If using a 60 gtt set, that would be (100×60/60)=100 drops/min. (In practice, infusion pumps are often used for precise rates, but manual drip counting can be done by adjusting the roller clamp to achieve the needed drop rate.)
Besides drop factor, IV sets come in various lengths and gauge of tubing. Typical infant or “micro-injection” sets may be shorter or smaller-diameter for small volumes; standard adult sets are ~150–200 cm long. Blood transfusion sets usually have large-bore tubing and integrated 170–200 μm filters (and sometimes no drip chamber, using a different flow regulator), allowing faster flow without hemolysis. Some special sets include burette chambers (for measuring small volumes, often 150 mL maximum) or extension sets for accessing multiple lines (Y-connectors or stopcocks) during multi-drug infusions.
Setup and Use (Techniques)
Preparation: Wash hands and use gloves. Select an infusion set appropriate to the task (microdrip for low-rate infusions, macrodrip or pump tubing for high-rate). Inspect the sterile package and expiration date. Inspect the tubing and components for damage or leaks. Don’t touch the spike or patient connector tips with bare hands.
Priming (filling): Close the roller clamp, remove the cap, and insert the spike into the fluid bag or bottle port (for vented containers, the spike often has a built-in air vent; if not, a vented spike or filter may be used). Squeeze the drip chamber till it’s half-full. Then open the clamp slowly to allow fluid to fill the entire tubing, expelling all air; do not reclose until all air is out (air embolism risk). Ensure the drip chamber has fluid above the filter (if present) and all connectors are fluid-primed.
Hanging and connecting: Hang the fluid bag high (≥18″ above patient) to ensure gravity flow. Clean the IV catheter hub on the patient with antiseptic (“scrub the hub”) and allow to dry. Attach the tubing’s Luer connector firmly to the catheter hub (or needleless valve).
Adjusting flow: Unlock or open the roller clamp. Count the drops in the drip chamber to set the rate: count drops for 30–60 seconds and adjust the clamp to match the calculated drops/minute. If using an infusion pump, program the volume and rate (pump tubing has specific characteristics so use the manufacturer’s instructions). Monitor the drip: count and recalibrate as needed to maintain desired flow.
Medication injection: Many IV sets have Y-injection ports with self-sealing needleless access or rubber septa. When giving a bolus med, clean the port with antiseptic and attach a syringe, push in the drug, then flush with saline. Never inject directly into air (always check that a drop comes out or that catheter is patent). If multiple medications run, use multi-port manifolds or stopcocks as needed, flushing between drugs.
Site Maintenance: Periodically check the IV site and tubing: ensure no kinks, the drip chamber and filter are not clogged, and fluid is dripping. Confirm the roller clamp position (no unintended top-ups or free flow). Many protocols recommend changing out IV tubing (except during continuous blood products or fats) no more frequently than every 72–96 hours to minimize infection risk (always follow hospital policy).
Stopping infusion and disposal: When done, clamp the tubing before disconnecting to avoid blood backflow. Disconnect the tubing from the catheter (keeping the patient host end sterile until capped). Discard the entire set (spike, tubing, drip chamber, etc.) as sharps/biomedical waste in a puncture-proof container. Under infection-control guidelines, no part of the infusion set is reused or shared between patients. Always wear gloves and maintain asepsis: as the CDC notes, all IV delivery equipment must be handled with sterile technique to prevent contamination.
Summary – Key Points
- Definition: IV (infusion) sets are sterile tubing assemblies for delivering fluids/medicines intravenously. They include a spike, drip chamber, tubing, clamps, ports, and a patient connector.
- Sizes/Drop Factor: Sets are color-coded/marked by drop factor: macrodrip (≈10–20 gtt/mL) for high rates, microdrip (60 gtt/mL) for precise low-rate infusions. Choose based on required flow.
- Flow rates: Calculate drops/min = (mL to infuse × drop factor) / infusion time (min). Use roller clamp to adjust until drops count is correct. Pumps add precision and alarms.
- Technique: Always prime tubing to remove air, connect using aseptic technique, clamp and unclamp to regulate flow, and flush lines before/after medication. Maintain site patency and check drip regularly.
- Safety: Use a new sterile set for each patient; dispose properly. Follow aseptic protocols to avoid air or microbial contamination. For example, CDC guidelines explicitly require that IV equipment remain sterile and contamination-free.
-
 Sale 10%
Original price $ 2.20Current price $ 1.98
Sale 10%
Original price $ 2.20Current price $ 1.98IV Sets, IV Administration Set 60 Drop Microdrip, Luer Lock Connector, 78 inch length
28 reviewsIV Set 60-Drop (Microdrip) A 60-drop microdrip IV set is a sterile infusion tubing kit used to deliver fluids or medications intravenously with ver...
View full details -
 Sale 22%
Original price $ 899.95Current price $ 699.95
Sale 22%
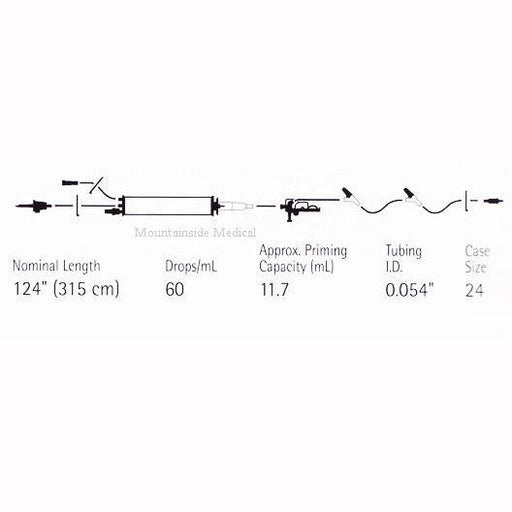
Original price $ 899.95Current price $ 699.95LifeShield PlumSet Microdrip SoluSet 60 Drop Iv Set
No reviewsLifeShield® PlumSet® Microdrip® SoluSet® 150 x 60 with small bore reduced priming volume tubing, convertible piercing pin, upper slide clamp, bact...
View full details -
 Sale 20%
Original price $ 479.95Current price $ 386.00
Sale 20%
Original price $ 479.95Current price $ 386.00Hospira LifeShield 14270-28 Burette Set
No reviewsThe Hospira LifeShield 14270-28 Burette Set, Macrobore, Clave Y-Site is comprised of a convertible piercing pin, three side clamps, a float valve, ...
View full details
People Also Searched For

IV Sets
IV sets (administration sets) are sterile, flexible plastic tubing systems used to deliver fluids, medications, or blood products directly into a patient’s vein. Key components include a spike for the bag, a drip chamber, a flow regulator (clamp), and a Luer connector. They are crucial for maintaining precise fluid administration and preventing air from entering the blood stream.
An IV set (or administration set) is a sterile, single-use plastic assembly that connects a fluid source (IV bag or bottle, blood unit, etc.) to a patient’s intravenous catheter or needle. The basic components include a rigid spike (to puncture the IV bag port), an air-vent or filter, a clear drip chamber, flexible tubing with clamps or flow regulators, one or more injection ports (often needleless) for meds, and a Luer-lock (or slip) connector at the patient end. For example, parts are typically named “spike, air vent cap, drip chamber (macro or micro), fluid filter, flow regulator (roller clamp or pinch clamp), injection site (Y-site with needle-free connector), and Luer connector”. Some sets also include built-in filters (e.g. 0.2 μm) or burette chambers for precise dosing. IV sets are always packaged sterile and used once – after priming and use they are discarded as medical sharps to prevent contamination. Proper aseptic technique is essential: all connection points (spike, ports, catheter) must remain sterile to avoid introducing pathogens. (Indeed, infection-control guidelines emphasize that IV delivery systems are sterile single-use devices and strict asepsis must be maintained during setup and use.
Sizes and Flow (Drop Factors)
IV tubing comes in various “drop factors” that define how many drops make 1 mL of fluid. Common designs are:
- Macrodrip (10–20 gtt/mL) – This is a larger-bore tubing where 1 mL ≈ 10–20 drops. Macrodrip sets are used for adult infusions or rapid fluid replacement (e.g. large-volume resuscitation). For example, a 15 gtt/mL set means ~15 drops = 1 mL.
- Microdrip (60 gtt/mL) – This fine-bore tubing yields 60 drops = 1 mL (each drop ≈0.017 mL), allowing very precise control. Microdrip sets are typically used for low-rate infusions (pediatrics, neonates, critical care) where accuracy matters.
Because microdrip sets produce tiny droplets, they are best when infusion rates are slow (commonly < 50–100 mL/hr) so that counting drops is feasible; macrodrip sets are preferred for higher rates (e.g. 100–2000 mL/hr) since fewer drops per mL can be counted or delivered more rapidly. To calculate flow:
- Drops per minute = (Volume (mL) to infuse × Drop factor (gtt/mL)) ÷ Time (min).
For instance, infusing 100 mL over 1 hour with a 15 gtt/mL set: (100 mL × 15 gtt/mL) ÷ 60 min ≈ 25 drops/min. If using a 60 gtt set, that would be (100×60/60)=100 drops/min. (In practice, infusion pumps are often used for precise rates, but manual drip counting can be done by adjusting the roller clamp to achieve the needed drop rate.)
Besides drop factor, IV sets come in various lengths and gauge of tubing. Typical infant or “micro-injection” sets may be shorter or smaller-diameter for small volumes; standard adult sets are ~150–200 cm long. Blood transfusion sets usually have large-bore tubing and integrated 170–200 μm filters (and sometimes no drip chamber, using a different flow regulator), allowing faster flow without hemolysis. Some special sets include burette chambers (for measuring small volumes, often 150 mL maximum) or extension sets for accessing multiple lines (Y-connectors or stopcocks) during multi-drug infusions.
Setup and Use (Techniques)
Preparation: Wash hands and use gloves. Select an infusion set appropriate to the task (microdrip for low-rate infusions, macrodrip or pump tubing for high-rate). Inspect the sterile package and expiration date. Inspect the tubing and components for damage or leaks. Don’t touch the spike or patient connector tips with bare hands.
Priming (filling): Close the roller clamp, remove the cap, and insert the spike into the fluid bag or bottle port (for vented containers, the spike often has a built-in air vent; if not, a vented spike or filter may be used). Squeeze the drip chamber till it’s half-full. Then open the clamp slowly to allow fluid to fill the entire tubing, expelling all air; do not reclose until all air is out (air embolism risk). Ensure the drip chamber has fluid above the filter (if present) and all connectors are fluid-primed.
Hanging and connecting: Hang the fluid bag high (≥18″ above patient) to ensure gravity flow. Clean the IV catheter hub on the patient with antiseptic (“scrub the hub”) and allow to dry. Attach the tubing’s Luer connector firmly to the catheter hub (or needleless valve).
Adjusting flow: Unlock or open the roller clamp. Count the drops in the drip chamber to set the rate: count drops for 30–60 seconds and adjust the clamp to match the calculated drops/minute. If using an infusion pump, program the volume and rate (pump tubing has specific characteristics so use the manufacturer’s instructions). Monitor the drip: count and recalibrate as needed to maintain desired flow.
Medication injection: Many IV sets have Y-injection ports with self-sealing needleless access or rubber septa. When giving a bolus med, clean the port with antiseptic and attach a syringe, push in the drug, then flush with saline. Never inject directly into air (always check that a drop comes out or that catheter is patent). If multiple medications run, use multi-port manifolds or stopcocks as needed, flushing between drugs.
Site Maintenance: Periodically check the IV site and tubing: ensure no kinks, the drip chamber and filter are not clogged, and fluid is dripping. Confirm the roller clamp position (no unintended top-ups or free flow). Many protocols recommend changing out IV tubing (except during continuous blood products or fats) no more frequently than every 72–96 hours to minimize infection risk (always follow hospital policy).
Stopping infusion and disposal: When done, clamp the tubing before disconnecting to avoid blood backflow. Disconnect the tubing from the catheter (keeping the patient host end sterile until capped). Discard the entire set (spike, tubing, drip chamber, etc.) as sharps/biomedical waste in a puncture-proof container. Under infection-control guidelines, no part of the infusion set is reused or shared between patients. Always wear gloves and maintain asepsis: as the CDC notes, all IV delivery equipment must be handled with sterile technique to prevent contamination.
Summary – Key Points
- Definition: IV (infusion) sets are sterile tubing assemblies for delivering fluids/medicines intravenously. They include a spike, drip chamber, tubing, clamps, ports, and a patient connector.
- Sizes/Drop Factor: Sets are color-coded/marked by drop factor: macrodrip (≈10–20 gtt/mL) for high rates, microdrip (60 gtt/mL) for precise low-rate infusions. Choose based on required flow.
- Flow rates: Calculate drops/min = (mL to infuse × drop factor) / infusion time (min). Use roller clamp to adjust until drops count is correct. Pumps add precision and alarms.
- Technique: Always prime tubing to remove air, connect using aseptic technique, clamp and unclamp to regulate flow, and flush lines before/after medication. Maintain site patency and check drip regularly.
- Safety: Use a new sterile set for each patient; dispose properly. Follow aseptic protocols to avoid air or microbial contamination. For example, CDC guidelines explicitly require that IV equipment remain sterile and contamination-free.



