Filters
- Connectors (1)
- Needles Connectors (1)
- Needles Ports (1)
- SwabCap (1)
- SwabCaps (1)
- 3M Curos (2)
- Curos Caps (1)
- Disinfectant Cleaner (2)
- Disinfecting Caps (1)
- Medical Connector (2)
- Medisca (2)
- Needless caps (1)
- Port Cleaner (2)
- Port Disinfectant (2)
- Luer Replacement Caps (1)
- Needless Caps (1)
- Red Caps (1)
- braun (1)
- Braun 418020 (1)
- Intermittent (1)
- Intermittent Injection Cap (1)
- IV Cap (1)
- IV Injection Cap (1)
- IV Therapy (1)
- IV Therapy Management (1)
- Curos Jet (1)
- Disinfectant Caps (1)
Needleless IV Connector Disinfectant Caps
-
 Sale 21%
Original price $ 0.95Current price $ 0.75
Sale 21%
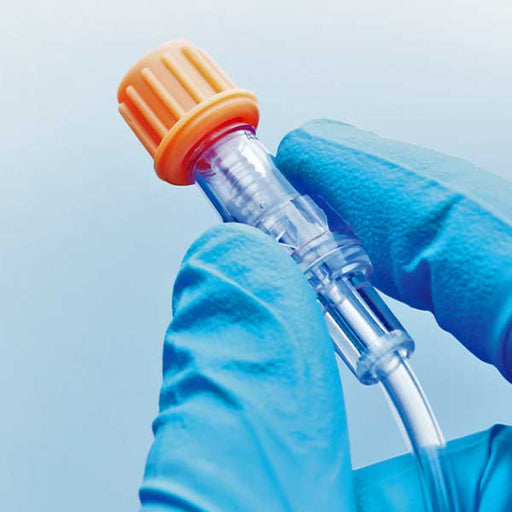
Original price $ 0.95Current price $ 0.75SwabCap Disinfectant Caps for Needles Connector Ports SCXT3
ICU MedicalNo reviewsSwabCap is a disinfectant cap designed to be placed over the needle-free connector port of medical devices, such as IV lines or IV sets, to prevent...
View full details -
 Sale 21%
Original price $ 175.00Current price $ 139.00
Sale 21%
Original price $ 175.00Current price $ 139.003M Curos IV Needleless Connector Disinfecting Caps 270/Box
3M HealthcareNo reviews3M Curos IV Needleless Connector Disinfecting Caps are medical devices designed to disinfect needleless connectors on intravenous (IV) tubing. Thes...
View full details -
 Sale
from $ 0.39
Sale
from $ 0.39B Braun Red Caps, Replacement Caps for IV Administration Sets
B BraunNo reviewsB Braun Red Caps are replacement caps designed specifically for IV administration sets, which are used to deliver medications or fluids directly in...
View full details -
 Sale 37%
Original price $ 29.95Current price $ 18.80
Sale 37%
Original price $ 29.95Current price $ 18.80Braun Intermittent Injection Cap 418020, 10/pk
BraunNo reviewsThe Braun Intermittent Injection Cap is a medical device used in intravenous therapy. It serves as a connector for IV lines, allowing medications o...
View full details -
 Sale 18%
Original price $ 195.00Current price $ 159.95
Sale 18%
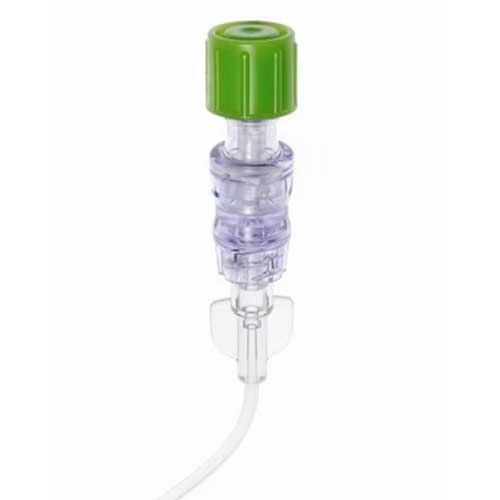
Original price $ 195.00Current price $ 159.953M Curos Jet Disinfecting Caps for Needleless Port Connectors CFJ1 Series 270/Box
3M HealthcareNo reviews3M Curos Jet Disinfecting Caps for Needleless Port Connectors CFJ1 Series are single-use, disposable caps designed to disinfect and protect needlel...
View full details
- Product Purpose: Needleless IV connector disinfectant caps prevent contamination and reduce bloodstream infection risk by applying antimicrobial solutions to IV connection points.
- Primary Users: Hospitals, clinics, ambulatory care centres, EMS teams, medspas, dental practices, veterinary clinics, and institutional healthcare buyers.
- Collection Scope: Multiple cap types compatible with different IV systems, connector styles, and clinical protocols; alcohol-based and alternative antimicrobial formulations.
- Clinical Standard: Products support infection control objectives outlined by CDC, APIC, and facility accreditation requirements including Joint Commission standards.
- Ordering Model: Wholesale bulk pricing with volume discounts; licensed healthcare professionals eligible for purchase; 5% off first order for new customers.
- Shipping & Support: Free US shipping on orders over $100; dedicated sales support for bulk orders and account management via phone or email.
- Standard Needleless Connector Caps: Designed for standard Luer lock and slip-tip connectors; available in multiple sizes for central lines, PICC lines, and peripheral access.
- Alcohol-Based Disinfecting Caps: Contain 70% isopropyl alcohol formulation; 15–30 second contact time for pathogen elimination; rapid evaporation after application.
- Injection Caps: Smaller-profile caps for low-volume injection ports and specialized IV systems; compatible with varied connector types.
- Color-Coded Cap Systems: Available in standardized colors to support visual identification and protocol adherence within facility workflows.
- Bulk Packaging Options: Individual sterile units, boxes, and case quantities with wholesale pricing; customizable order volumes for facility inventory management.
- Specialty Caps: Available for PICC lines, femoral catheters, and other specialized access points; compatible with manufacturer-specific connector designs.
| Cap Type | Antimicrobial Agent | Contact Time | Best For | Connector Compatibility |
|---|---|---|---|---|
| Standard Disinfecting Cap | 70% Isopropyl Alcohol | 15–30 seconds | Central lines, PICC lines, routine IV access | Luer lock, slip-tip needleless connectors |
| Injection Cap | 70% Isopropyl Alcohol | 15–30 seconds | Low-volume injection ports, specialty IV systems | Smaller Luer connectors, female ports |
| PICC Line Cap | 70% Isopropyl Alcohol | 15–30 seconds | Peripherally inserted central catheters | PICC-specific connectors |
| Clave Cap | 70% Isopropyl Alcohol | 15–30 seconds | Clave-compatible connectors | Clave male Luer connectors |
- Hospital and Acute Care Settings: Central line maintenance, PICC line management, and routine IV access protection in inpatient units and intensive care environments.
- Ambulatory Care and Clinics: Infusion clinics, dialysis centers, and routine outpatient IV therapy requiring consistent infection prevention between uses.
- EMS and Emergency Response: Field IV lines and rapid-access ports requiring protection during transport and between interventions.
- MedSpa and Aesthetic Practices: Injectable administration and IV therapy protocols for cosmetic and wellness procedures.
- Dental and Oral Surgery: IV sedation lines and local anesthetic delivery requiring connector protection between injections.
- Veterinary and Animal Care: IV access protection in animal treatment and surgical protocols.
- Home Health and Visiting Nurse Services: PICC line and peripheral IV maintenance in patient homes between clinical visits.
Clinical Use Note: This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. All usage must follow facility infection control protocols and manufacturer instructions. It is not a substitute for professional medical advice, diagnosis, or treatment.
- Identify Your Connector Type: Determine whether your facility uses Luer lock, slip-tip, Clave, or specialized needleless connectors. Cap compatibility is essential for proper fit and function.
- Assess Line Placement and Duration: Central lines and PICC lines benefit most from regular disinfection; peripheral IV lines and short-term access may require different cap specifications. Select based on anticipated dwell time.
- Consider Contact Time Requirements: Most alcohol-based caps require 15–30 seconds of contact time for optimal antimicrobial effect. Verify your facility's protocol aligns with product specifications.
- Evaluate Packaging and Volume Needs: Calculate monthly consumption based on facility size and IV procedure volume. Bulk packaging offers cost savings and ensures consistent supply.
- Review Color-Coding Standards: If your facility uses color-coded systems for line identification or protocol adherence, select cap colors that integrate with existing workflows.
- Confirm Regulatory Alignment: Verify that selected products meet your facility's accreditation standards and infection control policy requirements. Joint Commission and other oversight bodies provide guidance.
- Consult Infection Prevention Team: Work with your facility's infection preventionist or clinical leadership to ensure product selection supports institutional protocols and clinical objectives.
- Wholesale Pricing: Direct-supplier pricing available for licensed healthcare professionals, clinics, hospitals, EMS teams, medspas, dental practices, and institutional buyers. Volume discounts apply to bulk orders.
- First-Time Customer Discount: New customers receive 5% off their first purchase. Account setup requires valid medical license or institutional credentials verification.
- Free Shipping: Orders over $100 qualify for free standard US shipping. Expedited shipping options available for time-sensitive orders.
- Bulk Order Support: Dedicated sales team available to support facility procurement, volume negotiations, and supply chain planning. Contact +1 (888) 687-4334 or sales@mountainside-medical.com.
- Licensing and Compliance: Product purchase may require valid medical license or institutional healthcare provider status. License verification completed during account setup.
- Inventory Management: Account holders can track order history, manage reorders, and access historical pricing. Flexible ordering allows one-time purchases or scheduled recurring supply.
- Customer Support: Technical questions about product compatibility, usage, or ordering? Our healthcare supply specialists are available by phone or email during business hours.
Medical Device Disclaimer:
Needleless IV connector disinfectant caps are medical devices intended for use by licensed healthcare professionals or under the direct guidance of a qualified medical provider. These products are designed to support infection control measures and are not substitutes for comprehensive sterile technique, proper IV maintenance protocols, or professional medical care.
Product Use: All products must be used in accordance with manufacturer instructions for use, facility infection control policies, and applicable regulatory guidelines. Improper use, storage, or application may compromise product efficacy and patient safety.
Clinical Effectiveness: While these products are designed to reduce contamination risk at IV connection points, their effectiveness depends on correct application technique, appropriate contact time with the antimicrobial solution, and integration into comprehensive infection prevention protocols. Results vary based on facility protocols, user technique, and clinical environment.
Regulatory Status: Products in this collection are cleared or approved for sale as medical devices under FDA regulations. Specific regulatory clearance numbers are available upon request or in individual product documentation.
Not for Self-Treatment: These products are not appropriate for self-administration or use outside of professional healthcare settings. Patients should not attempt to apply or manage disinfecting caps without professional guidance.
Storage and Shelf Life: Products must be stored according to manufacturer specifications. Check product packaging for sterility assurance period and expiration dates. Do not use products beyond their expiration date.
Questions or Concerns: If you have questions about product suitability for your facility's specific needs, contact our sales team or consult with your facility's infection prevention specialist.
-
What is the difference between needleless connector caps and disinfecting caps?
Needleless connector caps provide physical protection for IV connectors, while disinfecting caps actively apply antimicrobial solutions to eliminate pathogens on connector surfaces. Disinfecting caps serve a dual function: mechanical coverage plus chemical disinfection. Many healthcare facilities use disinfecting caps specifically because of their infection prevention benefits beyond basic protection.
-
How long should disinfecting caps remain in contact with the connector?
Most alcohol-based disinfecting caps require 15–30 seconds of contact time for effective antimicrobial action against common healthcare pathogens. Always follow the manufacturer's specific contact time recommendations, as duration may vary between products. Facility protocols should standardize this timing to ensure consistent application.
-
Are these caps compatible with all needleless connectors?
No. Disinfecting caps are designed for specific connector types including Luer lock, slip-tip, and Clave systems. Verify your facility's connector type before ordering. Our product categories and comparison chart detail compatibility information. If you're uncertain about connector compatibility, contact our sales team at +1 (888) 687-4334.
-
Can these caps be used on peripheral IV lines or only central lines?
Disinfecting caps are appropriate for both central lines and peripheral IV lines. However, their primary infection prevention benefit is most significant for longer-dwelling lines such as central lines, PICC lines, and ports with higher infection risk. Product selection depends on your facility's protocols and the specific line type.
-
What antimicrobial agents are used in these caps?
Most products in this collection use 70% isopropyl alcohol as the primary antimicrobial agent. Alcohol-based formulations are effective against bacteria, fungi, and many viruses within the specified contact time. Alternative formulations may be available; consult individual product specifications or contact our sales team for options.
-
How should disinfecting caps be stored?
Store caps according to manufacturer specifications, typically in a cool, dry location away from direct sunlight. Maintain sterile packaging integrity until use. Check product labeling for specific storage temperature ranges and shelf-life information. Do not use caps beyond their expiration date.
-
Are there any compatibility issues with specific IV medications or fluids?
Alcohol-based disinfecting caps are compatible with most IV medications and fluids. However, if your facility uses specialized medications or compounds, verify compatibility with the specific cap product. Some facilities may have protocol restrictions; consult your infection prevention team or the product manufacturer if you have concerns.
-
Do I need a medical license to purchase these products?
Yes. Needleless IV connector disinfectant caps are medical devices intended for use by licensed healthcare professionals. Account setup requires verification of valid medical license or institutional healthcare provider credentials. New customers should contact our sales team at +1 (888) 687-4334 or sales@mountainside-medical.com to complete licensing verification.
-
What bulk ordering options are available?
Mountainside Medical supports facility bulk purchasing with volume discounts. Order quantities can be customized based on your facility's needs. For bulk orders, pricing negotiations, and account management, contact our dedicated sales team at +1 (888) 687-4334 or sales@mountainside-medical.com.
-
How quickly can I receive an order?
Free US shipping applies to orders over $100 with standard delivery. Expedited shipping options are available for time-sensitive orders. Contact our sales team for specific delivery timeframes based on your location and product availability.
-
Do you offer samples or trial quantities?
Not specified in provided data. Contact our sales team at +1 (888) 687-4334 or sales@mountainside-medical.com to discuss sample availability, trial quantities, or special requests for new facility accounts.
-
What support is available if I have questions about product selection?
Our healthcare supply specialists are available to answer questions about product compatibility, infection control applications, and ordering. Reach out by phone at +1 (888) 687-4334 or email sales@mountainside-medical.com during business hours. You can also consult with your facility's infection prevention team for clinical guidance on product selection.
- Product Purpose: Needleless IV connector disinfectant caps prevent contamination and reduce bloodstream infection risk by applying antimicrobial solutions to IV connection points.
- Primary Users: Hospitals, clinics, ambulatory care centres, EMS teams, medspas, dental practices, veterinary clinics, and institutional healthcare buyers.
- Collection Scope: Multiple cap types compatible with different IV systems, connector styles, and clinical protocols; alcohol-based and alternative antimicrobial formulations.
- Clinical Standard: Products support infection control objectives outlined by CDC, APIC, and facility accreditation requirements including Joint Commission standards.
- Ordering Model: Wholesale bulk pricing with volume discounts; licensed healthcare professionals eligible for purchase; 5% off first order for new customers.
- Shipping & Support: Free US shipping on orders over $100; dedicated sales support for bulk orders and account management via phone or email.
- Standard Needleless Connector Caps: Designed for standard Luer lock and slip-tip connectors; available in multiple sizes for central lines, PICC lines, and peripheral access.
- Alcohol-Based Disinfecting Caps: Contain 70% isopropyl alcohol formulation; 15–30 second contact time for pathogen elimination; rapid evaporation after application.
- Injection Caps: Smaller-profile caps for low-volume injection ports and specialized IV systems; compatible with varied connector types.
- Color-Coded Cap Systems: Available in standardized colors to support visual identification and protocol adherence within facility workflows.
- Bulk Packaging Options: Individual sterile units, boxes, and case quantities with wholesale pricing; customizable order volumes for facility inventory management.
- Specialty Caps: Available for PICC lines, femoral catheters, and other specialized access points; compatible with manufacturer-specific connector designs.
| Cap Type | Antimicrobial Agent | Contact Time | Best For | Connector Compatibility |
|---|---|---|---|---|
| Standard Disinfecting Cap | 70% Isopropyl Alcohol | 15–30 seconds | Central lines, PICC lines, routine IV access | Luer lock, slip-tip needleless connectors |
| Injection Cap | 70% Isopropyl Alcohol | 15–30 seconds | Low-volume injection ports, specialty IV systems | Smaller Luer connectors, female ports |
| PICC Line Cap | 70% Isopropyl Alcohol | 15–30 seconds | Peripherally inserted central catheters | PICC-specific connectors |
| Clave Cap | 70% Isopropyl Alcohol | 15–30 seconds | Clave-compatible connectors | Clave male Luer connectors |
- Hospital and Acute Care Settings: Central line maintenance, PICC line management, and routine IV access protection in inpatient units and intensive care environments.
- Ambulatory Care and Clinics: Infusion clinics, dialysis centers, and routine outpatient IV therapy requiring consistent infection prevention between uses.
- EMS and Emergency Response: Field IV lines and rapid-access ports requiring protection during transport and between interventions.
- MedSpa and Aesthetic Practices: Injectable administration and IV therapy protocols for cosmetic and wellness procedures.
- Dental and Oral Surgery: IV sedation lines and local anesthetic delivery requiring connector protection between injections.
- Veterinary and Animal Care: IV access protection in animal treatment and surgical protocols.
- Home Health and Visiting Nurse Services: PICC line and peripheral IV maintenance in patient homes between clinical visits.
Clinical Use Note: This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. All usage must follow facility infection control protocols and manufacturer instructions. It is not a substitute for professional medical advice, diagnosis, or treatment.
- Identify Your Connector Type: Determine whether your facility uses Luer lock, slip-tip, Clave, or specialized needleless connectors. Cap compatibility is essential for proper fit and function.
- Assess Line Placement and Duration: Central lines and PICC lines benefit most from regular disinfection; peripheral IV lines and short-term access may require different cap specifications. Select based on anticipated dwell time.
- Consider Contact Time Requirements: Most alcohol-based caps require 15–30 seconds of contact time for optimal antimicrobial effect. Verify your facility's protocol aligns with product specifications.
- Evaluate Packaging and Volume Needs: Calculate monthly consumption based on facility size and IV procedure volume. Bulk packaging offers cost savings and ensures consistent supply.
- Review Color-Coding Standards: If your facility uses color-coded systems for line identification or protocol adherence, select cap colors that integrate with existing workflows.
- Confirm Regulatory Alignment: Verify that selected products meet your facility's accreditation standards and infection control policy requirements. Joint Commission and other oversight bodies provide guidance.
- Consult Infection Prevention Team: Work with your facility's infection preventionist or clinical leadership to ensure product selection supports institutional protocols and clinical objectives.
- Wholesale Pricing: Direct-supplier pricing available for licensed healthcare professionals, clinics, hospitals, EMS teams, medspas, dental practices, and institutional buyers. Volume discounts apply to bulk orders.
- First-Time Customer Discount: New customers receive 5% off their first purchase. Account setup requires valid medical license or institutional credentials verification.
- Free Shipping: Orders over $100 qualify for free standard US shipping. Expedited shipping options available for time-sensitive orders.
- Bulk Order Support: Dedicated sales team available to support facility procurement, volume negotiations, and supply chain planning. Contact +1 (888) 687-4334 or sales@mountainside-medical.com.
- Licensing and Compliance: Product purchase may require valid medical license or institutional healthcare provider status. License verification completed during account setup.
- Inventory Management: Account holders can track order history, manage reorders, and access historical pricing. Flexible ordering allows one-time purchases or scheduled recurring supply.
- Customer Support: Technical questions about product compatibility, usage, or ordering? Our healthcare supply specialists are available by phone or email during business hours.
Medical Device Disclaimer:
Needleless IV connector disinfectant caps are medical devices intended for use by licensed healthcare professionals or under the direct guidance of a qualified medical provider. These products are designed to support infection control measures and are not substitutes for comprehensive sterile technique, proper IV maintenance protocols, or professional medical care.
Product Use: All products must be used in accordance with manufacturer instructions for use, facility infection control policies, and applicable regulatory guidelines. Improper use, storage, or application may compromise product efficacy and patient safety.
Clinical Effectiveness: While these products are designed to reduce contamination risk at IV connection points, their effectiveness depends on correct application technique, appropriate contact time with the antimicrobial solution, and integration into comprehensive infection prevention protocols. Results vary based on facility protocols, user technique, and clinical environment.
Regulatory Status: Products in this collection are cleared or approved for sale as medical devices under FDA regulations. Specific regulatory clearance numbers are available upon request or in individual product documentation.
Not for Self-Treatment: These products are not appropriate for self-administration or use outside of professional healthcare settings. Patients should not attempt to apply or manage disinfecting caps without professional guidance.
Storage and Shelf Life: Products must be stored according to manufacturer specifications. Check product packaging for sterility assurance period and expiration dates. Do not use products beyond their expiration date.
Questions or Concerns: If you have questions about product suitability for your facility's specific needs, contact our sales team or consult with your facility's infection prevention specialist.
-
What is the difference between needleless connector caps and disinfecting caps?
Needleless connector caps provide physical protection for IV connectors, while disinfecting caps actively apply antimicrobial solutions to eliminate pathogens on connector surfaces. Disinfecting caps serve a dual function: mechanical coverage plus chemical disinfection. Many healthcare facilities use disinfecting caps specifically because of their infection prevention benefits beyond basic protection.
-
How long should disinfecting caps remain in contact with the connector?
Most alcohol-based disinfecting caps require 15–30 seconds of contact time for effective antimicrobial action against common healthcare pathogens. Always follow the manufacturer's specific contact time recommendations, as duration may vary between products. Facility protocols should standardize this timing to ensure consistent application.
-
Are these caps compatible with all needleless connectors?
No. Disinfecting caps are designed for specific connector types including Luer lock, slip-tip, and Clave systems. Verify your facility's connector type before ordering. Our product categories and comparison chart detail compatibility information. If you're uncertain about connector compatibility, contact our sales team at +1 (888) 687-4334.
-
Can these caps be used on peripheral IV lines or only central lines?
Disinfecting caps are appropriate for both central lines and peripheral IV lines. However, their primary infection prevention benefit is most significant for longer-dwelling lines such as central lines, PICC lines, and ports with higher infection risk. Product selection depends on your facility's protocols and the specific line type.
-
What antimicrobial agents are used in these caps?
Most products in this collection use 70% isopropyl alcohol as the primary antimicrobial agent. Alcohol-based formulations are effective against bacteria, fungi, and many viruses within the specified contact time. Alternative formulations may be available; consult individual product specifications or contact our sales team for options.
-
How should disinfecting caps be stored?
Store caps according to manufacturer specifications, typically in a cool, dry location away from direct sunlight. Maintain sterile packaging integrity until use. Check product labeling for specific storage temperature ranges and shelf-life information. Do not use caps beyond their expiration date.
-
Are there any compatibility issues with specific IV medications or fluids?
Alcohol-based disinfecting caps are compatible with most IV medications and fluids. However, if your facility uses specialized medications or compounds, verify compatibility with the specific cap product. Some facilities may have protocol restrictions; consult your infection prevention team or the product manufacturer if you have concerns.
-
Do I need a medical license to purchase these products?
Yes. Needleless IV connector disinfectant caps are medical devices intended for use by licensed healthcare professionals. Account setup requires verification of valid medical license or institutional healthcare provider credentials. New customers should contact our sales team at +1 (888) 687-4334 or sales@mountainside-medical.com to complete licensing verification.
-
What bulk ordering options are available?
Mountainside Medical supports facility bulk purchasing with volume discounts. Order quantities can be customized based on your facility's needs. For bulk orders, pricing negotiations, and account management, contact our dedicated sales team at +1 (888) 687-4334 or sales@mountainside-medical.com.
-
How quickly can I receive an order?
Free US shipping applies to orders over $100 with standard delivery. Expedited shipping options are available for time-sensitive orders. Contact our sales team for specific delivery timeframes based on your location and product availability.
-
Do you offer samples or trial quantities?
Not specified in provided data. Contact our sales team at +1 (888) 687-4334 or sales@mountainside-medical.com to discuss sample availability, trial quantities, or special requests for new facility accounts.
-
What support is available if I have questions about product selection?
Our healthcare supply specialists are available to answer questions about product compatibility, infection control applications, and ordering. Reach out by phone at +1 (888) 687-4334 or email sales@mountainside-medical.com during business hours. You can also consult with your facility's infection prevention team for clinical guidance on product selection.





