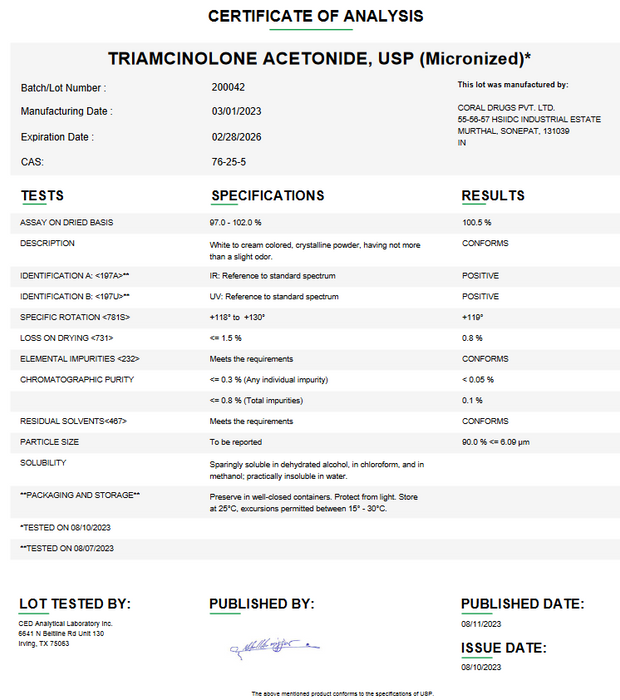
Triamcinolone Acetonide (Micronized) API Powder
Triamcinolone acetonide is a potent synthetic corticosteroid (glucocorticoid) used to reduce inflammation. As an API (active pharmaceutical ingredient) powder, it meets United States Pharmacopeia standards and is used by pharmacists to compound prescription medications. The drug is a more powerful analogue of prednisone (about eight times more potent) and is the active steroid in products like Kenalog (injectable), Aristocort, and topical preparations. In appearance, USP-grade triamcinolone acetonide (micronized) is a white to cream-colored crystalline powder with only a slight odor. Its chemical formula is C₂₄H₃₁FO₆ (molecular weight ≈434.5). The powder is micronized (grind to very fine particles) so that at least 90% of particles are below 10 µm in size. This fine particle size allows uniform dispersion in creams or injectable suspensions and can improve bioavailability when applied topically. Triamcinolone acetonide powder is virtually insoluble in water and only sparingly soluble in alcohol, so formulations typically suspend or emulsify the drug rather than dissolve it fully.
Triamcinolone Acetonide (micronized) api powder is a white to cream colored, crystalline powder, having not more than a slight odor. It is sparingly soluble in dehydrated alcohol and practically insoluble in water. Particles micronized so that 90% are less than 10μm.
Key Properties (API powder): White/cream crystalline powder with slight odor; USP grade meets pharmacopeial standards (high purity). Chemical formula C₂₄H₃₁FO₆ (MW 434.5). Micronized to ≤10 µm sizing for consistency. Physically, it’s practically insoluble in water (suspensions used). Being a corticosteroid, it is potent: about 8× the potency of prednisone.
Triamcinolone Acetonide USP (Micronized) for Compounding (API) is an active pharmaceutical ingredient used to create customized medications for treating inflammatory conditions. As a potent corticosteroid, it helps reduce inflammation, redness, and swelling in conditions such as eczema, psoriasis, and allergic reactions. Micronization enhances its absorption and efficacy, allowing for precise formulation tailored to patient-specific needs. Compounding pharmacists can adjust the dosage and delivery form, offering targeted relief and improved management of dermatological and localized inflammatory disorders.
Triamcinolone Acetonide USP is an active pharmaceutical ingredient (API) used in certain medications. It is a synthetic corticosteroid that works by reducing inflammation in the body. It is used to treat a variety of conditions including allergies, skin disorders, and respiratory diseases. Triamcinolone Acetonide USP is available in various forms including creams, ointments, and injections.
Triamcinolone Acetonide USP is a synthetic corticosteroid medication used in compounding to treat inflammation, itching, and redness associated with certain skin conditions. It can also be used to treat joint pain and inflammation caused by certain types of arthritis. In compounding, this medication is often combined with other ingredients to create customized topical, oral, or injectable formulations as directed by a healthcare provider.
Pharmaceutical Uses
s a USP API, micronized triamcinolone acetonide is used to compound anti-inflammatory medications. Compounding pharmacists incorporate it into topical formulations (creams, ointments, gels) and injectable suspensions or depot injections for localized or systemic treatment of inflammation. For example, it is the active ingredient in cortisone creams for eczema or dermatitis and in injectable preparations for joint or soft-tissue inflammation. It can also be used in nasal spray or dental paste formulations. Typical compound strengths range from low percentages in creams (e.g. 0.02–0.1%) up to full injection doses (40–80 mg per mL for intramuscular/intra-articular injections). Because the raw API is extremely potent, dosage and dilution are carefully controlled. For injections, the micronized powder is suspended in a sterile vehicle (often with preservatives like benzyl alcohol) and must be shaken before use to re-suspend the steroid crystals. For transdermal use, it is mixed into a cream or ointment base; the fine powder ensures even distribution and smooth texture.
-
Applications: Treating inflammatory and allergic conditions. Compounded creams/ointments for eczema, psoriasis, contact dermatitis, and rashes (as a high-potency topical steroid). Injectable suspensions (e.g. Kenalog-40/80 equivalents) for acute joint flares, bursitis, or severe allergic reactions. Intralesional injections (skin lesions) and off-label uses (e.g. ocular or intratympanic injections) under specialist guidance. Also used in nasal sprays or inhalers for allergic rhinitis/asthma in its CFC-free formulations (e.g. Nasacort). Because it is not water soluble, all these formulations rely on suspensions, emulsions or solutions with suitable carriers.
Handling and Storage: Being a potent steroid API, triamcinolone acetonide powder must be handled with care. Personal Protection: Wear gloves, eye protection, and protective clothing when handling the powder. Avoid inhalation or ingestion – the powder is labeled “Harmful if swallowed” and may cause skin or eye irritation on contact. Work in a well-ventilated area or fume hood. Storage: Store the powder in a tightly closed container to protect it from moisture and light. Keep it locked or in a secure area if required (per safety guidelines), out of reach of patients and unqualified personnel. Room temperature storage (20–25 °C) is typical unless otherwise specified. Stability and Expiry: Follow the manufacturer’s expiration date and USP stability data. Since it is a crystalline powder, it is relatively stable, but should be kept dry to avoid degradation. During compounding, use aseptic technique especially if preparing injectables. The USP monograph for triamcinolone acetonide (as part of finished products) typically requires content uniformity (90–110% of label claim), and compounding pharmacists should verify potency when possible (e.g. with assay kits or certified titration) to ensure each batch meets specifications.
-
Precautions: Because it is an immunosuppressive steroid, minimize exposure. Do not use API powder in neonates (injections of triamcinolone often contain benzyl alcohol, which is contraindicated in infants). Avoid contamination: use a new sterile syringe/gauge for injectable compounding and never reuse the powder for multiple patients. Dispose of any unused opened powder according to hazardous drug waste guidelines (it’s a Category 4 anticancer-equivalent drug in USP chapter K3 caution). In case of contact, wash skin thoroughly; if inhaled or ingested, seek medical attention. Triamcinolone (like all corticosteroids) can suppress adrenal function if absorbed systemically, so compounded doses should be accurate and minimal effective (USP-labelled dosage forms cap therapeutic ranges).
Triamcinolone acetonide USP (micronized) is a high-potency corticosteroid active ingredient supplied as a fine white/cream powder. It is intended for use by pharmacists to compound anti-inflammatory medications – for example, into creams for skin conditions or suspensions for injection. The micronized particle size (<10 µm) ensures even dispersion in formulations. Being a strength-calibrated USP powder, it must be measured and stored properly. Wear appropriate PPE (gloves/eye protection) during handling and store the container tightly sealed. With correct formulation techniques, triamcinolone acetonide API allows providers to create tailor-made steroid medications that deliver potent localized therapy for inflammation and allergy.
CAS Number: 76-25-5
Property: Light Sensitive