Filters
- 0.9% benzyl alcohol (1)
- Aesthetic Supplies MedSpa Supplies & Cosmetic Surgery Supplies (1)
- Bac (1)
- bac water (1)
- Bacterio (1)
- bacteriostatic (1)
- bacteriostatic water (1)
- Diluent (4)
- dilution (1)
- doctor-only (10)
- Hospira Pfizer (4)
- Multiple dose Sterile water (1)
- Multiple Dose Vial (1)
- reconstitution (1)
- Sale (1)
- Static water (1)
- sterile water (1)
- Sterile Water For Injection (8)
- sterile water injection (2)
- water for diluting (2)
- water for injection (6)
- Hospira Vials (2)
- Injectable (1)
- Injection Vials (2)
- Sterile water 20 ml (1)
- Sterile water Injection (3)
- 50 ml (1)
- Intramuscular (1)
- Intravenous (3)
- Single-use (1)
- Sterle Water for Injection (1)
- Glass Vials (1)
- Single Dose (2)
- Sterile Vials (1)
- Sterile Water 100 ml (1)
- 10 ml (1)
- 10 ml Sterile water (1)
- Sterile Water 10 ml (1)
- Sterile Water Injection (1)
- Sterile Water 5 mL (1)
- Bag (1)
- bags (1)
- Dehydration (1)
- Hydration (1)
- IV (1)
- IV Bag (2)
- Iv Bags (1)
- Iv Solution (1)
- IV Sterile Water (1)
- IV Water (1)
- Rehydration (1)
- Severe Dehydration (1)
- Water IV (1)
- flush syringes (1)
- prefilled syringe (1)
- prefilled syringes (1)
- Syringes (1)
- Diluentant (1)
- Single Dose Vial (1)
- Infusion Therapy (1)
Bacteriostatic Water and Sterile Water for Injection
Bacteriostatic Water
Bacteriostatic Water for Injection is sterile, non-pyrogenic water that contains 0.9% benzyl alcohol as a preservative. It is intended only for injection use – specifically as a diluent or solvent to dissolve or dilute injectable medications. The benzyl alcohol acts as a bacteriostatic agent, meaning it inhibits (but does not kill) bacterial growth in the solution. This preservative property allows a single vial to be used for multiple withdrawals (i.e. multi-dose) under sterile technique. (By contrast, plain sterile water for injection contains no preservative and is meant for single-use only.)
Key Features
- Composition: Bacteriostatic water contains 0.9% (9 mg/mL) benzyl alcohol in USP-grade sterile water. This matches official guidelines (for example, Hospira’s package insert).
- Purpose: It is used to reconstitute dry (lyophilized) drugs or dilute concentrated injectable drugs according to the drug manufacturer’s instructions. For example, many powdered antibiotics, hormones (like peptides), or vaccines require a diluent to prepare a dose. Healthcare providers draw the needed amount of bacteriostatic water to dissolve the medication before injecting it intravenously (IV), intramuscularly (IM), or subcutaneously (SC).
- Multi-dose Vial: Because of the benzyl alcohol preservative, bacteriostatic water typically comes in larger vials (commonly 30 mL plastic vials) that remain sterile for multiple uses. Each time a dose is withdrawn with a sterile needle, the benzyl alcohol prevents microbial contamination from multiplying in the vial. (If opened and stored properly, the vial can often be used for days or weeks – usually up to 28 days – until the expiration date, as long as sterility is maintained.)
- Not a Medication By Itself: Bacteriostatic water has no pharmacological effect other than serving as a vehicle. It does not contain electrolytes or other drugs, and it will not treat any condition on its own.
Uses and Applications
- Drug Reconstitution: Commonly, bacteriostatic water is used to prepare injections of medications that come as dry powders or highly concentrated solutions. The amount used depends on the drug’s guidelines. For instance, a vial of peptide or vaccine may instruct “add 2 mL of sterile water” – in such cases bacteriostatic water can be used if suitable.
- Dilution for Injection: It can also dilute liquid concentrates (such as certain hormone or fertility drugs) to the correct dose before injection. Once mixed, the final solution should be approximately isotonic if given IV (meaning it should contain appropriate solute concentration for safety).
- Multi-route: After reconstitution, the resulting mixture can be administered IV, IM, or SC per the drug’s instructions. (However, note that when injecting intramuscularly or subcutaneously, any trace preservative is generally well tolerated; the main caution is with IV use, as below.)
- Not for Drinking or Large-volume IV: Bacteriostatic water is only for injectable pharmaceuticals, not for hydration or oral use. It should never be administered intravenously by itself, because it contains no salts and is hypotonic. IV injection of plain water – even with a little benzyl alcohol – can cause red blood cells to swell and burst (hemolysis). Similarly, it’s not meant for large-volume IV infusion.
Safety and Precautions
- Aseptic Technique: Always use sterile gloves and needles when withdrawing from the vial. Bacteriostatic vials allow multiple entries, but each entry must be done with a new sterile syringe/needle to avoid introducing pathogens. Discard the vial when its expiration or recommended in-use period is reached.
- Not for Neonates: Because benzyl alcohol can be toxic to very young infants (it has been linked to “gasping syndrome” and other serious effects), bacteriostatic water must never be used for neonates or newborn medications. In those cases, preservative-free sterile water for injection (without benzyl alcohol) is required.
- Allergies: Patients with a known allergy to benzyl alcohol or similar preservatives should not receive medications reconstituted with bacteriostatic water.
- Label Warnings: The official product label explicitly warns not to inject bacteriostatic water by itself into the bloodstream. It also notes that it is for diluting or dissolving drugs per their instructions – meaning you should only use it if the medication specifically allows it. Some medications (especially certain biologicals or vaccines) require saline or preservative-free water, so always follow the drug manufacturer’s guidance.
- Environmental Conditions: Store unopened vials at controlled room temperature (typically 20–25 °C) as directed. Do not freeze. After opening, keep the rubber stopper clean and recap it. If you see any cloudiness, discoloration, or particulates in the water, do not use it.
In summary: Bacteriostatic water is essentially sterile water for injection with 0.9% benzyl alcohol added. It is used as a diluent to reconstitute or dilute injectable medications, allowing multi-dose use thanks to its preservative. It is not a drug by itself and must be used exactly as instructed (with sterile technique and appropriate medications). Because it contains benzyl alcohol, it is contraindicated in newborns and should not be injected into the bloodstream alone
-
 Sale 20%
Original price $ 379.00Current price $ 305.00
Sale 20%
Original price $ 379.00Current price $ 305.00Bacteriostatic Water Injection by Hospira, Multiple Dose Vials 30 mL 25/Pack (Rx)
1115 reviewsBacteriostatic Water for Injection is a pharmaceutical-grade sterile diluent formulated with 0.9% benzyl alcohol as a preservative, enabling safe m...
View full details🔒 Medical License Required -
 Sale 20%
Original price $ 275.00Current price $ 219.00
Sale 20%
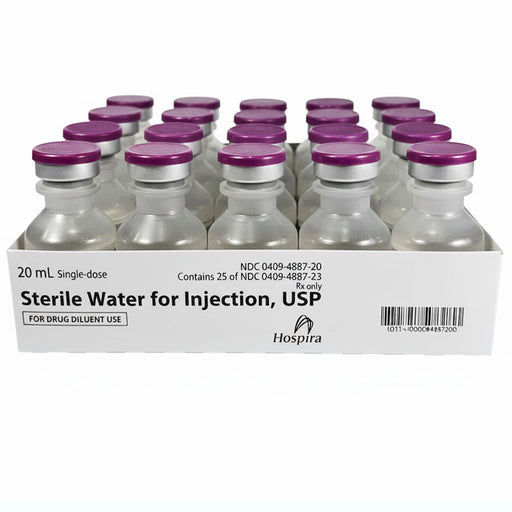
Original price $ 275.00Current price $ 219.00Sterile Water Injection 20 mL Single-Dose Vials by Hospira 25/Tray (Rx)
54 reviewsSterile Water for Injection 20 mL by Pfizer is a pharmaceutical-grade solvent used to dilute or dissolve medications for injection. It is employed ...
View full details🔒 Medical License Required -
 Sale 15%
Original price $ 385.00Current price $ 329.00
Sale 15%
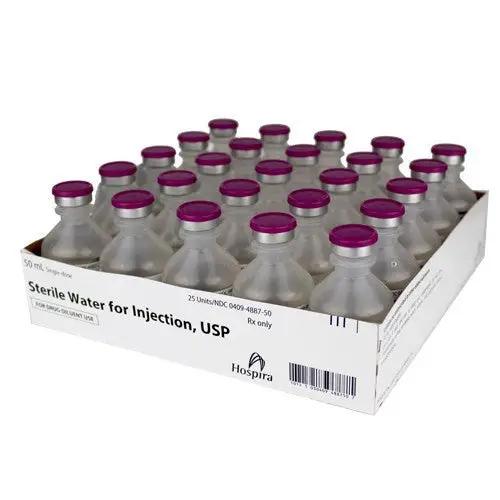
Original price $ 385.00Current price $ 329.00Sterile Water for Injection 50 mL Single-Use Vials, 25/Tray (Rx)
30 reviewsSterile Water for Injection 50 mL Single-Use Vials are used as a solvent or diluent for the preparation of medications for injection. These vials p...
View full details🔒 Medical License Required -
 Sale 15%
Original price $ 445.00Current price $ 379.00
Sale 15%
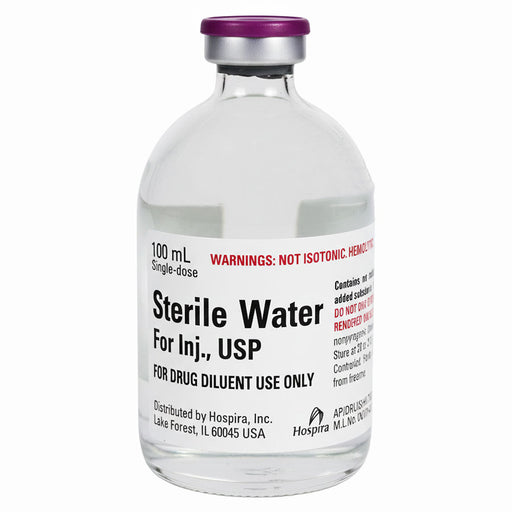
Original price $ 445.00Current price $ 379.00Sterile Water for Injection 100 mL Glass Vials, 25/Tray (Rx)
1 reviewSterile Water for Injection 100 mL Glass Vials are used as a solvent or diluent for the preparation of medications intended for injection. This ste...
View full details🔒 Medical License Required -
 Sale 25%
Original price $ 219.95Current price $ 165.00
Sale 25%
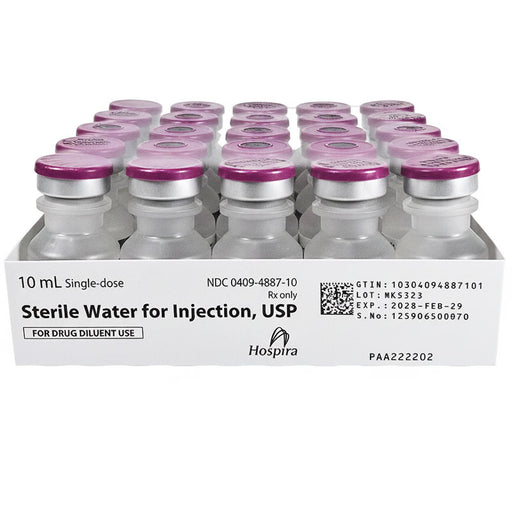
Original price $ 219.95Current price $ 165.00Sterile Water for Injection 10 ml Single-Dose Vials 25/Tray by Hospira (Rx)
3 reviewsHospira Sterile Water for Injection is a pharmaceutical-grade, non-pyrogenic sterile water intended for use as a solvent or diluent for the prepara...
View full details🔒 Medical License Required -
 Sale 5%
Original price $ 169.00Current price $ 159.99
Sale 5%
Original price $ 169.00Current price $ 159.99Sterile Water for Injection 5 mL Single Dose Vials, 25/Pack (Rx)
1 reviewSterile Water for Injection 5 mL is a sterile, single-use solution that is used to dilute and reconstitute medications for injection. It is water t...
View full details🔒 Medical License Required -
 Sale 35%
Original price $ 199.99Current price $ 129.00
Sale 35%
Original price $ 199.99Current price $ 129.00IV Bags, Sterile Water for Injection 1000 mL by B Braun (12/Case) (Rx)
2 reviewsSterile Water for Injection IV Bags 1000 mL by B. Braun is a pharmaceutical-grade, nonpyrogenic water meant for intravenous use, primarily as a sol...
View full details🔒 Medical License Required -
 Sale 36%
Original price $ 54.95Current price $ 35.00
Sale 36%
Original price $ 54.95Current price $ 35.00Sterile Water for Injection Prefilled Syringes 10 mL Single-Dose, 10/Box Medefil (Rx)
No reviewsMedefil Prefilled Sterile Water for Injection Syringes 10 mL. Medefil prefilled sterile water for injection syringes are designed for single use on...
View full details🔒 Medical License Required -
 Sale 16%
Original price $ 94.00Current price $ 78.60
Sale 16%
Original price $ 94.00Current price $ 78.60Sterile Water for Injection 10 mL Vials, 10/Pack (Rx)
No reviewsSterile Water for Injection is a clear, colorless, and sterile liquid used for medical purposes. It is made by purifying and sterilizing water, and...
View full details🔒 Medical License Required -
 Sale 24%
Original price $ 235.00Current price $ 179.00
Sale 24%
Original price $ 235.00Current price $ 179.00IV Bag Sterile Water for Injection Solution 1000ml, 12/Case (Rx)
11 reviewsSterile Water 1000mg IV bag used for intravenous use. Sterile water iv solution is a sterile, nonpyrogenie solution. Single-use container. Note: Th...
View full details🔒 Medical License Required
People Also Searched For
Bacteriostatic Water
Bacteriostatic Water for Injection is sterile, non-pyrogenic water that contains 0.9% benzyl alcohol as a preservative. It is intended only for injection use – specifically as a diluent or solvent to dissolve or dilute injectable medications. The benzyl alcohol acts as a bacteriostatic agent, meaning it inhibits (but does not kill) bacterial growth in the solution. This preservative property allows a single vial to be used for multiple withdrawals (i.e. multi-dose) under sterile technique. (By contrast, plain sterile water for injection contains no preservative and is meant for single-use only.)
Key Features
- Composition: Bacteriostatic water contains 0.9% (9 mg/mL) benzyl alcohol in USP-grade sterile water. This matches official guidelines (for example, Hospira’s package insert).
- Purpose: It is used to reconstitute dry (lyophilized) drugs or dilute concentrated injectable drugs according to the drug manufacturer’s instructions. For example, many powdered antibiotics, hormones (like peptides), or vaccines require a diluent to prepare a dose. Healthcare providers draw the needed amount of bacteriostatic water to dissolve the medication before injecting it intravenously (IV), intramuscularly (IM), or subcutaneously (SC).
- Multi-dose Vial: Because of the benzyl alcohol preservative, bacteriostatic water typically comes in larger vials (commonly 30 mL plastic vials) that remain sterile for multiple uses. Each time a dose is withdrawn with a sterile needle, the benzyl alcohol prevents microbial contamination from multiplying in the vial. (If opened and stored properly, the vial can often be used for days or weeks – usually up to 28 days – until the expiration date, as long as sterility is maintained.)
- Not a Medication By Itself: Bacteriostatic water has no pharmacological effect other than serving as a vehicle. It does not contain electrolytes or other drugs, and it will not treat any condition on its own.
Uses and Applications
- Drug Reconstitution: Commonly, bacteriostatic water is used to prepare injections of medications that come as dry powders or highly concentrated solutions. The amount used depends on the drug’s guidelines. For instance, a vial of peptide or vaccine may instruct “add 2 mL of sterile water” – in such cases bacteriostatic water can be used if suitable.
- Dilution for Injection: It can also dilute liquid concentrates (such as certain hormone or fertility drugs) to the correct dose before injection. Once mixed, the final solution should be approximately isotonic if given IV (meaning it should contain appropriate solute concentration for safety).
- Multi-route: After reconstitution, the resulting mixture can be administered IV, IM, or SC per the drug’s instructions. (However, note that when injecting intramuscularly or subcutaneously, any trace preservative is generally well tolerated; the main caution is with IV use, as below.)
- Not for Drinking or Large-volume IV: Bacteriostatic water is only for injectable pharmaceuticals, not for hydration or oral use. It should never be administered intravenously by itself, because it contains no salts and is hypotonic. IV injection of plain water – even with a little benzyl alcohol – can cause red blood cells to swell and burst (hemolysis). Similarly, it’s not meant for large-volume IV infusion.
Safety and Precautions
- Aseptic Technique: Always use sterile gloves and needles when withdrawing from the vial. Bacteriostatic vials allow multiple entries, but each entry must be done with a new sterile syringe/needle to avoid introducing pathogens. Discard the vial when its expiration or recommended in-use period is reached.
- Not for Neonates: Because benzyl alcohol can be toxic to very young infants (it has been linked to “gasping syndrome” and other serious effects), bacteriostatic water must never be used for neonates or newborn medications. In those cases, preservative-free sterile water for injection (without benzyl alcohol) is required.
- Allergies: Patients with a known allergy to benzyl alcohol or similar preservatives should not receive medications reconstituted with bacteriostatic water.
- Label Warnings: The official product label explicitly warns not to inject bacteriostatic water by itself into the bloodstream. It also notes that it is for diluting or dissolving drugs per their instructions – meaning you should only use it if the medication specifically allows it. Some medications (especially certain biologicals or vaccines) require saline or preservative-free water, so always follow the drug manufacturer’s guidance.
- Environmental Conditions: Store unopened vials at controlled room temperature (typically 20–25 °C) as directed. Do not freeze. After opening, keep the rubber stopper clean and recap it. If you see any cloudiness, discoloration, or particulates in the water, do not use it.
In summary: Bacteriostatic water is essentially sterile water for injection with 0.9% benzyl alcohol added. It is used as a diluent to reconstitute or dilute injectable medications, allowing multi-dose use thanks to its preservative. It is not a drug by itself and must be used exactly as instructed (with sterile technique and appropriate medications). Because it contains benzyl alcohol, it is contraindicated in newborns and should not be injected into the bloodstream alone










