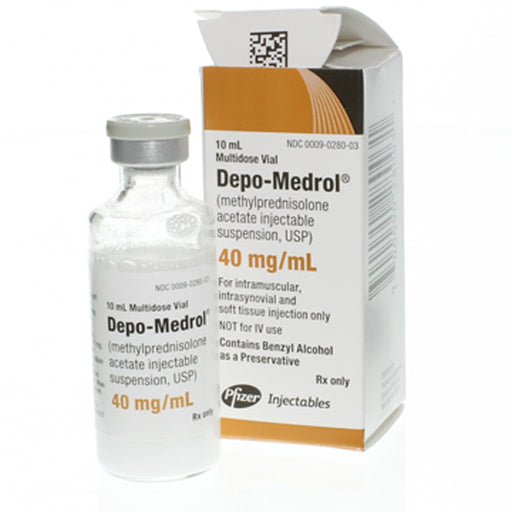
Depo-Medrol (Methylprednisolone Acetate) Injection
Depo-Medrol is a corticosteroid injection (methylprednisolone acetate) used to rapidly reduce inflammation. It is a potent glucocorticoid (synthetic cortisol analog) that suppresses the immune system and inflammatory pathways. Commonly used brand names include Depo-Medrol (Pfizer) and Medrol/Solu-Medrol (Pfizer generics). As an injectable suspension, Depo-Medrol is administered by a healthcare provider either intramuscularly (IM) for systemic effect or directly into a joint or tendon sheath (intra-articular/soft tissue) for local effect.
Mechanism of Action
-
Glucocorticoid effect: Methylprednisolone binds intracellular glucocorticoid receptors. It alters gene transcription to decrease production of inflammatory cytokines, prostaglandins, and leukotrienes, and it reduces migration of immune cells to inflamed tissue. The result is a broad anti-inflammatory and immunomodulatory effect (reducing swelling, redness, pain).
-
Depot suspension: The acetate salt is insoluble, so the drug remains at the injection site and is released slowly over weeks. (This contrasts with “Solu-Medrol” [methylprednisolone sodium succinate], which is water-soluble and acts more quickly for intravenous use.)
Uses
Depo-Medrol is used for inflammatory and immune-mediated conditions. Depending on the route, it can be used for systemic inflammatory diseases or for local joint/tissue inflammation:
-
Systemic (IM injection): Used when rapid anti-inflammatory effect is needed and oral therapy is not possible. Indications include severe asthma or allergy flares, drug reactions, autoimmune flare-ups (e.g. rheumatoid arthritis, systemic lupus), dermatologic flares (pemphigus, Stevens-Johnson), inflammatory bowel disease, and adrenal insufficiency (as part of replacement therapy). (The Mayo Clinic describes methylprednisolone injection as relieving inflammation in many organ systems: asthma, arthritis, skin conditions, lupus, ulcerative colitis, and more.)
-
Intra-articular or soft-tissue injection: Very common for local relief of joint and tendon inflammation. FDA-labeled uses (as adjunctive short-term therapy) include acute arthritis flares (gouty arthritis, rheumatoid arthritis, osteoarthritis with synovitis), bursitis, tenosynovitis (tendon sheath inflammation), and epicondylitis (tennis/golfer’s elbow). Injection into a swollen joint can rapidly reduce pain and stiffness. (Depo-Medrol is also injected into keloids, alopecia plaques, and other localized lesions, but these dermatologic uses are less common.)
In practice, Depo-Medrol injections are often performed for osteoarthritis knee flares, shoulder or hip bursitis, Achilles or patellar tendinitis, and the like. It is generally used as a short-term measure (“tide over”) during acute exacerbations or when adding steroid-sparing therapy. Chronic daily use is avoided if possible.
Administration and Dosage
Depo-Medrol comes as a sterile suspension (e.g. 40 mg/mL or 80 mg/mL vials) of methylprednisolone acetate. It must be given by injection using sterile technique. Because it is a single-dose vial, any unused portion should be discarded after injection.
-
Intramuscular injection: For systemic effect (e.g. severe asthma or allergic reaction), Depo-Medrol is injected deeply into a large muscle (usually the gluteus). Doses vary by condition, commonly in the range of 40–120 mg per injection (1–3 mL if 40 mg/mL). For example, a 80 mg IM dose might be given weekly or as needed. (MedicineNet notes typical Depo-Medrol IM doses of 10–80 mg every 1–2 weeks, though dosing is individualized.) The effect of one IM injection may last ~1–2 weeks.
-
Intra-articular/soft-tissue injection: The dose depends on the size of the joint or lesion. A general guideline (from FDA labeling) is: Large joints (knee, ankle, shoulder) – 20–80 mg (0.5–2 mL); Medium joints (elbow, wrist) – 10–40 mg (0.25–1 mL); Small joints (fingers, toes) – 4–10 mg (0.1–0.25 mL). Tendon sheath or bursa injections usually use 4–30 mg. For example, a knee might receive 40 mg once, while an inflamed finger joint might get 5 mg. Practitioners often aspirate synovial fluid first to confirm needle placement, then inject Depo-Medrol into the joint space. The joint is gently moved afterward to mix the steroid suspension. Repeat injections (if needed) are typically spaced by several weeks (e.g. ≥4–6 weeks) to monitor response and avoid excess steroid exposure.
As a depot, one injection deposits methylprednisolone for weeks of effect. However, because of side effects, therapy is generally limited to the shortest duration needed. (For example, steroid injections for osteoarthritis are usually repeated at most every few months, if at all.)
Side Effects and Adverse Reactions
Depo-Medrol has the same side effect profile as other corticosteroids, plus some local effects from injection. Patients often experience corticosteroid-related changes:
-
Immediate local effects: Pain or discomfort at the injection site is common. Rarely, a transient increase in pain (“steroid flare”) can occur over the next 24–48 hours after a Joint injection. If sterile technique is not perfect, there is a risk of local infection (septic arthritis, abscess). Local tissue effects include skin or subcutaneous fat thinning, depigmentation, or small depressions at the injection site. (Because steroids can suppress skin repair, skin atrophy or delayed wound healing is noted. To minimize this, the smallest effective volume is used and care is taken not to inject into the dermis.)
-
Metabolic/endocrine effects: Corticosteroids raise blood glucose and can worsen or precipitate diabetes. Patients frequently gain weight and retain fluid (edema). They may develop “moon face” and Cushingoid appearance. Blood pressure often increases. Long-term use suppresses the adrenal glands: suddenly stopping steroids can cause adrenal insufficiency. Corticosteroids also cause sodium retention and potassium loss (hypokalemia) in susceptible individuals.
-
Cardiovascular: Fluid retention and hypertension can stress the heart; patients with heart disease or high blood pressure should be monitored. In very rare instances, serious events like congestive heart failure, thromboembolism, or arrhythmias have been reported.
-
Neurologic/psych: Steroids can cause headache, dizziness, or intracranial pressure rise. Mood changes are common: euphoria, irritability, anxiety, insomnia, or depression. Rarely, “steroid psychosis” (mania or psychotic behavior) can occur with high doses. Because of this, patients should be monitored for severe mood swings.
-
Musculoskeletal: Prolonged use results in osteoporosis. (Even a few months of high-dose continuing steroid can cause bone loss.) Patients are advised to get adequate calcium/vitamin D, and periodic bone density monitoring is prudent. Avascular necrosis of bone (e.g. hip necrosis) and tendon ruptures are rare but serious complications. Muscle weakness and “steroid myopathy” may develop with chronic use.
-
Dermatologic: Acne, increased facial hair, and thinning of the scalp hair can occur. Fragile skin leads to easy bruising and petechiae. Steroid-induced striae (purple stretch marks) are possible.
-
Ocular: Long-term steroid use can lead to cataracts (posterior subcapsular cataracts) and glaucoma (increased intraocular pressure). Patients on chronic therapy should have periodic eye exams.
-
Gastrointestinal: Steroids increase appetite (often weight gain). They also increase stomach acid, raising the risk of peptic ulcers or GI bleeding, especially if combined with NSAIDs.
-
Immune/susceptibility: By dampening the immune response, steroids increase infection risk. Latent infections (tinea, herpes, tuberculosis) can flare. Patients on high-dose steroids should avoid exposure to contagious diseases (especially chickenpox or measles) because their symptoms may be blunted. Vaccinations: Live vaccines (e.g. MMR, varicella) are generally contraindicated while on immunosuppressive doses of steroids.
Summary of Common Reactions: MedicineNet notes that frequent side effects include fluid retention, weight gain, hypertension, low potassium, headache, muscle weakness, easy bruising, acne, glaucoma, cataracts, growth retardation in children, and psychiatric disturbances. These reflect systemic corticosteroid effects. In the short term (occasional injection), mild hypertension, hyperglycemia, and mood changes are often seen; in the long term (repeated injections), osteoporosis, adrenal suppression, and metabolic syndrome become concerns.
Precautions and Warnings
-
Injection technique: Depo-Medrol vials are single-use only. Do not mix the suspension with other drugs or diluents. Use a fresh sterile syringe/needle for each injection. When injecting into a joint, always aspirate synovial fluid first to confirm intra-articular placement. Avoid injecting into tendons (e.g. Achilles) directly, unless injecting the sheath, to prevent rupture.
-
Contraindications: Systemic fungal infections (IM use) and intrathecal/epidural administration are contraindicated.. Do not inject into unstable joints or joints without true synovial space (e.g. spinal facet joints). Avoid if severe infection is present near the injection site.
-
Diabetes/Hypertension: Use caution or adjust treatment in patients with diabetes, hypertension, or heart disease, as steroids can significantly worsen these conditions.
-
Adrenal insufficiency: In critically ill or stressed patients (trauma, surgery), steroid doses should be adjusted. Never stop prolonged therapy abruptly — taper gradually to avoid adrenal crisis.
-
Osteoporosis: Minimizing steroid exposure (dose and duration) is important to protect bones. Consider calcium/D supplements if prolonged use is needed.
-
Pediatrics: Growth suppression can occur in children. Do not exceed recommended pediatric doses without necessity. Growth should be monitored in any child on steroids.
-
Pregnancy/Breastfeeding: Corticosteroids can cause fetal harm in animals (cleft palate, etc). Use during pregnancy only if the benefit outweighs risk; infants born to mothers on steroids should be monitored for adrenal suppression. Methylprednisolone does appear in breast milk; brief use is unlikely to harm most infants, but caution is advised if nursing.
-
Drug interactions: Concurrent use with other immunosuppressants or NSAIDs can alter risks (e.g. more GI bleeding). Many drug interactions are possible with steroids (e.g. with anticoagulants, diabetes meds); clinicians adjust therapy accordingly.
Additional Notes
-
Duration of effect: One advantage of Depo-Medrol (acetate) over soluble steroids is prolonged action. A single injection can control symptoms for weeks, which can bridge the time until a slower-starting therapy (like DMARDs in RA) takes effect.
-
Generic availability: Depo-Medrol (brand by Pfizer) has generic equivalents (methylprednisolone acetate) sold by various companies. The term “Depo-Medrol” usually refers to the 40 mg/mL or 80 mg/mL methylprednisolone acetate suspension.
-
Related preparations: Do not confuse with Solu-Medrol (methylprednisolone sodium succinate), which is a water-soluble steroid often given IV. Solu-Medrol acts quickly (for acute crises), whereas the acetate (Depo-Medrol) acts slowly and persists.
-
Monitoring: Before and during therapy, doctors may check blood pressure, blood glucose, skin integrity, and bone density (if repeated use). Eye exams are recommended if steroids are used long-term.
Summary: Depo-Medrol is a powerful anti-inflammatory steroid injection used to quell severe joint and soft-tissue inflammation, and to manage other acute inflammatory conditions. It works by broadly suppressing the immune response. Patients often experience steroid-like side effects (fluid retention, high blood sugar, mood changes, etc.). Proper technique and cautious use (lowest effective dose, shortest duration) are essential to minimize risks. All use should follow medical guidance and the official prescribing information.