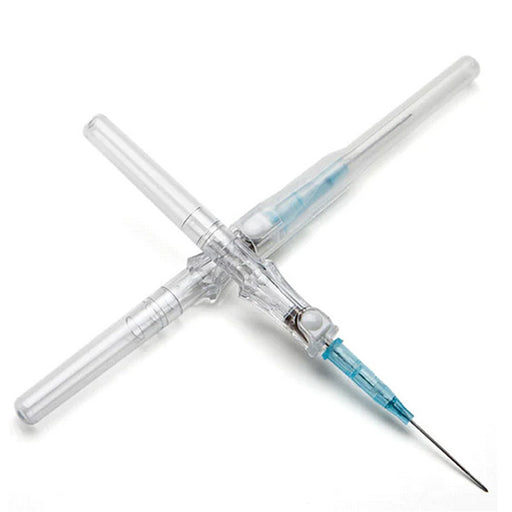
BD Nexiva Closed IV Catheter Needle with Sliding Safety is a cutting-edge medical device that combines the expertise of needle technology with a revolutionary safety mechanism. This catheter needle is designed to provide the most secure and painless intravenous access for patients while reducing the risk of needlestick injuries for healthcare professionals. The sliding safety feature allows for a single-handed activation, making it easy to use in emergency situations. The closed design ensures that the needle remains protected at all times and eliminates the need for additional capping devices, saving time and resources. With its advanced technology and user-friendly design, the Nexiva Closed IV Catheter Needle with Sliding Safety is a top choice for hospitals and medical facilities looking to improve patient care and the safety of their staff.
Benefits for BD Nexiva Closed IV Catheter Needle with Sliding Safety Shield
- Minimizes Risk of Needle Stick Injuries: The Nexiva Closed IV Catheter Needle with Sliding Safety Shield eliminates the need to recap a needle. This reduces the risk of needlestick injuries that can occur when recapping a needle.
- Minimizes Contamination Risk: The Nexiva Closed IV Catheter Needle features an internal closing lid that shields the needle while not in use. This reduces the risk of cross-contamination with other patients’ injection medications.
- Reduces Sharps Disposal Costs: Needles used with the Nexiva Closed IV Catheter Needle are discarded with the same sharps container system used for standard needle and syringe systems. This simplifies disposal processes and reduces the costs associated with dedicated medical waste pickup of sharps containers.
- Improves Patient Culture of Safety: The Nexiva Closed IV Catheter Needle and Shield helps hospitals and healthcare facilities foster a culture of safety amongst their staff and patients. The closed system and enhanced safety features of this device promote better protection against needle-stick injuries and cross contamination.
- Optimizes Efficiency in Use of Medicines: The Nexiva Closed IV Catheter Needle minimizes the amount of time medications are exposed to open air, reducing the possibility of spoilage or contamination. This extend the shelf life of medications and injectables and supports quality assurance and waste reduction initiatives in healthcare facilities.
- Standard adult peripheral IV access (20G): Routine medication delivery, IV hydration, and antibiotic infusion in adult patients with accessible forearm or hand veins.
- High-flow and transfusion access (18G): Indicated for blood product administration and faster-rate infusions where flow volume is a clinical consideration.
- Rapid fluid resuscitation (16G): Large-bore access for trauma, haemorrhage, or emergency volume replacement scenarios.
- Paediatric and geriatric access (22G–24G): Smaller gauge options intended to support IV access in patients with small, fragile, or difficult-to-cannulate peripheral veins.
- Neonatal access (24G): Fine-gauge option for neonatal and infant IV access in high-dependency or NICU settings.
- High-insertion-volume environments: The pre-assembled closed system is intended for settings where insertion frequency and blood exposure risk are both elevated — emergency departments, infusion centres, ICUs.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
The BD Nexiva Closed IV Catheter System is a sterile, single-use medical device intended exclusively for use by trained and licensed healthcare professionals. All insertion procedures must be performed in accordance with the manufacturer's official Instructions for Use (IFU), your facility's established IV access protocols, and applicable clinical guidelines.
The clinical information provided on this page — including gauge selection guidance, insertion overview, and system component descriptions — is intended as general reference for licensed healthcare procurement and clinical personnel only. It does not constitute medical advice and is not a substitute for professional clinical training, institutional protocols, or manufacturer IFU documentation.
Mountainside Medical does not manufacture this product. All product claims, specifications, and performance data referenced on this page reflect information provided by Becton, Dickinson and Company (BD Medical). Mountainside Medical makes no independent warranty regarding clinical performance outcomes.
Purchase of this product requires a valid US medical license. By purchasing, the buyer confirms they are a licensed healthcare professional or authorised institutional procurement agent operating within the scope of applicable federal and state regulations.
For complete safety, contraindication, and usage information, refer to the BD Nexiva product IFU available from Becton, Dickinson and Company.