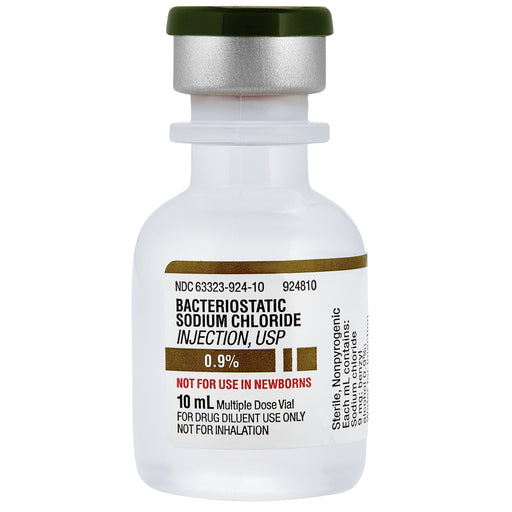
Bacteriostatic Sodium Chloride 0.9% Injection is a sterile normal saline solution (0.9% sodium chloride in water) that contains a small amount of a bacteriostatic preservative. In most formulations, the preservative is benzyl alcohol (0.9% benzyl alcohol per mL), which inhibits the growth of bacteria. The presence of this preservative keeps the solution sterile between uses, allowing the product to be packaged in a multiple-dose vial for repeated withdrawals without contamination. In other words, “bacteriostatic” means it prevents bacteria from growing, so the same vial of saline can be used multiple times (within its recommended timeframe) instead of being single-use.
Composition and Properties
This injection is essentially 0.9% sodium chloride (NaCl) dissolved in sterile water—with 9 mg of sodium chloride per milliliter, making it isotonic with blood and tissues. The isotonic nature means it has the same salt concentration as normal body fluids, so it won’t cause cells to shrink or swell when injected. In addition to salt, each milliliter contains about 9 mg of benzyl alcohol (0.9%) as the bacteriostatic agent. The benzyl alcohol does not treat infection, but its job is to keep the solution sterile by inhibiting bacterial growth if the vial is used more than once. The solution is labeled “nonpyrogenic”, meaning it’s free from substances that cause fever, and it’s intended for parenteral use (injection). Because it’s a USP-grade preparation, it meets quality standards for purity and sterility required for injectable solutions.
Intended Use (Diluent for Medications)
Bacteriostatic 0.9% Sodium Chloride Injection is not a medication on its own – it’s a diluent or solvent used to prepare other drugs for injection. According to its labeling, this solution is indicated only for diluting or dissolving drugs for injection (whether intravenous (IV), intramuscular (IM), or subcutaneous (SC)). For example, if you have a vial of powdered medication that needs to be injected, you might add this bacteriostatic saline to reconstitute (dissolve) the powder into a liquid form for injection. Similarly, it can be used to dilute concentrated drugs to the proper strength before injecting them. Healthcare providers also sometimes use sterile saline solutions to flush IV lines or injection sites, but in many cases preservative-free saline is preferred for flushing (especially in sensitive patients). The key point is that Bacteriostatic Sodium Chloride 0.9% Injection provides a sterile salt-water medium for mixing medications, and its bacteriostatic property allows the vial to be used for multiple doses or multiple patients (with proper sterile technique) instead of being discarded after a single use.
Uses
-
Diluting or dissolving drugs for injection.
-
Flushing IV lines (less common).
-
Used in situations when multiple-dose vials are required, because the bacteriostatic agent prevents bacterial contamination during repeated entries.
Order Bacteriostatic Sodium Chloride 0.9% Injection 30 mL x 25/Pack by Pfizer Today!
NDC: 0409-1966-07 | UPC: 304091966024 Fast Shipping | Bulk Pack | FDA-Approved | Trusted by Hospitals Everywhere
Provide your clinical team with the safest, most reliable multi-dose injectable saline available on the market—choose Pfizer Bacteriostatic Sodium Chloride for every reconstitution, dilution, and flush need.
Uses & Applications
Bacteriostatic Sodium Chloride 0.9% Injection is indicated for:
- Reconstitution and dilution of injectable medications: Antibiotics, biologics, hormones, and other drugs requiring sterile saline
- Flushing IV lines and catheters: To prevent blockage and maintain line patency
- Sterile wound irrigation in clinical settings
- Intravenous, intramuscular (IM), or subcutaneous (SC) injection as a vehicle for compatible drugs
- Preparing syringes for allergy shots, HRT, botulinum toxin, and other compounded injections
- Basic clinical saline needs where repeated withdrawals from a single vial are necessary
Instructions on How to Use
- Inspect vial: Ensure solution is clear with no particulates or discolouration.
- Sanitise vial stopper: Wipe with alcohol before each needle entry.
- Withdraw desired amount: Use a sterile syringe and needle for each access.
- Use strictly aseptic technique to avoid contamination.
- Inject, reconstitute, or flush as directed by accompanying medication, protocol, or provider’s instructions.
- Discard remaining solution 28 days after initial entry or sooner if contamination is suspected.
Not for direct IV infusion without proper mixing with medications unless specifically indicated.
Dosage and Administration
- For reconstitution/dilution:
- Follow medication-specific guidelines; use only the required volume for safe, effective mixing.
- For IV/IM/SC Injection as vehicle:
- Use as recommended by the medication manufacturer or the physician's protocol.
- For flushing catheters or IV lines:
- Typically 2–5 mL as needed, using sterile technique.
- Wound irrigation:
- Use per facility standard procedures.
NEVER use bacteriostatic saline in neonates or infants due to risk of benzyl alcohol toxicity (“gasping syndrome”).
Side Effects & Warnings
Common Side Effects:
- Mild injection site irritation, swelling, or redness, especially if injected IM/SC.
- Rarely, local allergic or site reactions due to benzyl alcohol preservative.
Serious Adverse Reactions:
- Hypersensitivity/anaphylactic reaction (rare)
- Hypernatremia or fluid overload from excessive administration (seen only in improper use)
- Neonatal gasping syndrome: NEVER use in infants; benzyl alcohol may be fatal.
Warnings:
- Not for use in infants <1 month or pregnant women unless specifically indicated/approved.
- Use in accordance with sterile procedure protocol; discard vial 28 days after first entry.
- If solution is cloudy or contains particulates, discard immediately.
- Overuse or inappropriate administration may result in electrolyte imbalance.
- Reconstitution of lyophilised injectables: Dissolves powdered medications (antibiotics, hormones, peptides) into solution for parenteral administration via IV, IM, or SC routes
- Dilution of concentrated injectables: Reduces concentrated drug solutions to the required administration strength prior to injection
- Peptide reconstitution: Widely used by licensed practitioners to reconstitute peptide-based compounds including GLP-1 receptor agonists (e.g. tirzepatide), growth hormone-related peptides (e.g. tesamorelin, retatrutide), and other research-designated injectable peptides
- Aesthetic and MedSpa applications: Used as a reconstitution medium for botulinum toxin products; the isotonic sodium chloride formulation is preferred by some injectors over hypotonic diluents for injection-site comfort considerations
- Multi-dose clinical workflow: The bacteriostatic preservative allows repeated vial entry under aseptic conditions — suitable for high-volume reconstitution settings including outpatient clinics, EMS formularies, and aesthetic practices
- Professional use only: Intended for use by licensed physicians, nurses, pharmacists, EMS professionals, dentists, veterinarians, and other qualified healthcare providers
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Bacteriostatic Sodium Chloride 0.9% Injection is a prescription-only (Rx) product. Purchase requires valid medical licensure. It is the responsibility of the purchasing professional to ensure compliance with all applicable federal, state, and local regulations governing the purchase, storage, handling, and administration of prescription pharmaceutical products.
This product contains benzyl alcohol 0.9% as a preservative. Benzyl alcohol is contraindicated in neonates and premature infants. This product is not indicated for large-volume parenteral administration or IV hydration.
Product information provided on this page references FDA-approved prescribing information (NDC 0409-1966-07) and USP compendial standards. Clinical decisions regarding product selection, reconstitution volumes, and administration must be made by a licensed healthcare professional in accordance with applicable clinical guidelines and facility protocols.
Mountainside Medical Equipment supplies this product for use by licensed healthcare professionals only. Content on this page is informational and does not constitute medical advice.