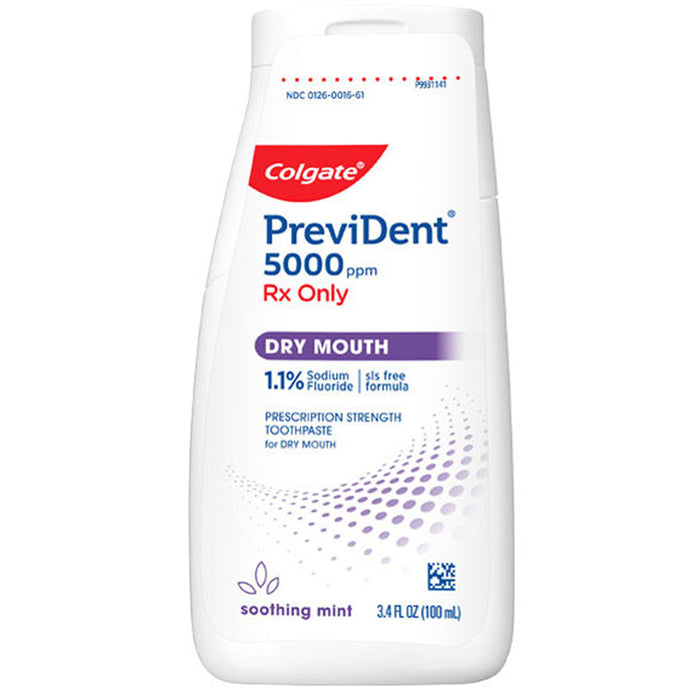
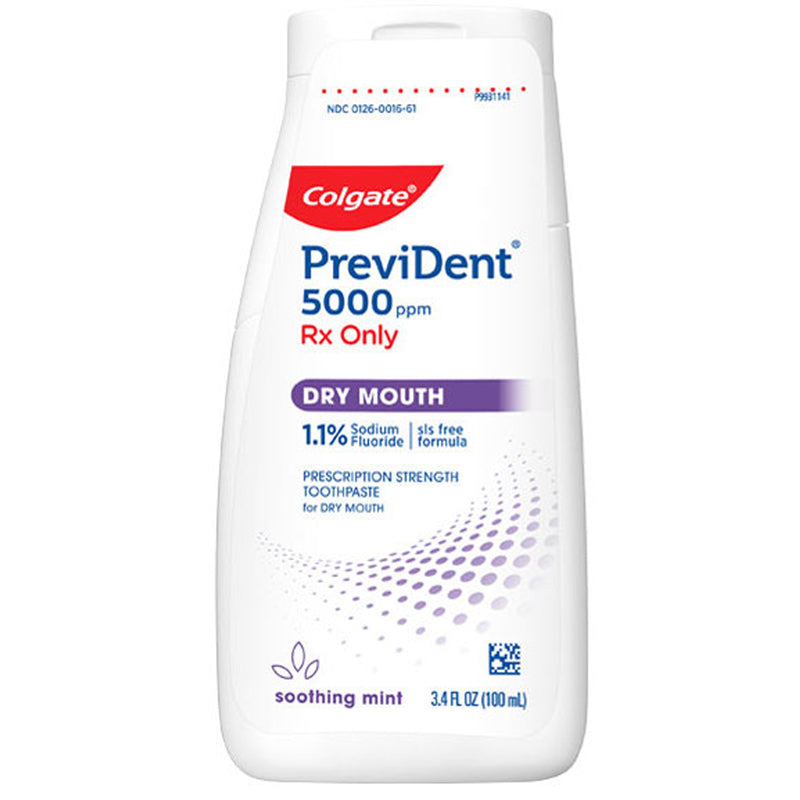
Colgate Prevident 5000 Dry Mouth Toothpaste Gel, 100mL
How to Order:
You will receive instructions on how to create an account along with Rx Ordering Details.
(Note: Acceptable licenses must have Prescriptive Authority in the license issuing state.)
Cart subtotal








 Sale 23%
Sale 23%
Colgate PreviDent 5000 Enamel Protect is designed help strengthening teeth and stop painful sensitivity. A self-topical neutral fluoride toothpaste...
View full details Sale 32%
Sale 32%
Colgate PreviDent 5000 Plus is a high fluoride toothpaste that delivers 4 to 5 times more fluoride than regular toothpaste and can help repair earl...
View full details Sale 30%
Sale 30%
Colgate PreviDent 5000 Brush-on Gel is a convenient sodium fluoride formula that’s perfect for patients requiring extra-strength protection for the...
View full details Sale 24%
Sale 24%
Colgate PreviDent Oral Rinse is a mint flavored dental rinse that targets root caries and tooth decay. Prevident oral rinse active ingredient: 0.2%...
View full details Sold out
Sold out
Chlorhexidine gluconate mouthwash is used to prevent and treat gum disease (gingivitis) and to control bacteria that can cause plaque in the mouth....
View full details Sale 33%
Sale 33%
SF 5000 Plus Sodium Fluoride 1.1% Dental Cream Spearmint Flavor is a prescription-strength toothpaste designed to prevent and protect against tooth...
View full details Sale 30%
Sale 30%
Prevident 5000 Sensitivity Relief Toothpaste is a neutral fluoride dentifrice oral paste containing 1.1% (w/w) sodium fluoride and 5% potassium ni...
View full details Sale 16%
Sale 16%
MI Paste is a topical dental product designed to restore and protect tooth enamel by providing essential minerals such as calcium and phosphate. It...
View full details Sale 24%
Sale 24%
Mi Paste Mint is a topical oral care cream used to remineralize and protect teeth, offering relief from tooth sensitivity and aiding in the prevent...
View full details Sale 16%
Sale 16%
MI Paste Plus is a topical tooth cream designed to strengthen and protect teeth by delivering essential minerals. It contains fluoride and Recalden...
View full details Sale
Sale
Dry Mouth Gel by GC America is a dental product that is used to help individuals who experience dry mouth symptoms. Dry mouth, also known as xerost...
View full details Sale 36%
Sale 36%
Dynarex Lemon Glycerin Swabsticks are a type of medical swabstick that is coated with a lemon-flavored glycerin solution. The lemon glycerin serves...
View full details Sale 31%
Sale 31%
Biotene dry mouth moisturizing relief spray is a saliva substitute used to provide relief for dry mouth symptoms. It helps to provide a lubricating...
View full details Sale 36%
Sale 36%
ACT Dry Mouth Relief Lozenges are a breakthrough product specifically designed to alleviate the discomfort of dry mouth. Dry mouth, or xerostomia, ...
View full details Sale
Sale
Biotene Dry Mouth Moisturizing Oral Rinse Mouthwash is a alcohol-free, sugar-free formula that soothes and moisturizes and helps you manage Dry Mou...
View full details Sale 47%
Sale 47%
Xylitol Dry Mouth Gum is a sugar free chewing gum that treats and relieves xerostomia (dry mouth). Xylitol Dry Mouth Gum is designed to help incre...
View full details Sale 28%
Sale 28%
Biotene Dry Mouth Treatment Fluoride Toothpaste in Fresh Mint is specifically formulated to alleviate dry mouth symptoms while promoting oral healt...
View full details Sale 28%
Sale 28%
XyliMelts help alleviate both dry mouth and bad breath symptoms. XyliMelts not only help increase saliva and freshen breath, but also last for hour...
View full details Sale 44%
Sale 44%
Hager Dry Mouth Drops Lozenges Cherry Flavor are an excellent solution for those experiencing dry mouth. This irritating condition, also known as x...
View full details Sale 15%
Sale 15%
Crest Gum Detoxify Toothpaste Deep Clean is a specialty dental hygiene product that helps to aid in the prevention of future cavities and reduce cu...
View full details Sale 25%
Sale 25%
Mi Paste Plus Mint is a topical oral cream designed to strengthen teeth and enhance overall dental health. It contains fluoride to aid in the remin...
View full details Sale 25%
Sale 25%
MI Paste with Recaldent is a dental product used to protect and strengthen tooth enamel by delivering essential minerals like calcium and phosphate...
View full details Sale 30%
Sale 30%
Mi Paste One is an advanced dental product that combines toothpaste and calcium/phosphate remineralization therapy in one convenient step. It is de...
View full details Sale
Sale
Dry Mouth Gel by GC America is a dental product that is used to help individuals who experience dry mouth symptoms. Dry mouth, also known as xerost...
View full details Sale 30%
Sale 30%
MI Paste® contains RECALDENT® (CPP-ACP); Casein Phosphopeptide (CPP) are natural occurring molecules which are able to release calcium and phosphat...
View full details Sale 23%
Sale 23%
Colgate PreviDent 5000 Enamel Protect is designed help strengthening teeth and stop painful sensitivity. A self-topical neutral fluoride toothpaste...
View full details Sale
Sale
Oral swabsticks, also known as Toothettes, are small medical devices used for oral care, typically made of soft foam or imitation cotton on a plast...
View full details Sale 23%
Sale 23%
Advanced Remineralizing Toothpaste for Ultimate Dental Health Unlock your brightest, healthiest smile with the leading anti-cavity solution recomme...
View full details Sale 31%
Sale 31%
MI Paste One Kids Anti-Cavity Toothpaste with Bubblegum Flavor is a toothpaste designed for children to help prevent cavities and maintain good ora...
View full details Sale 33%
Sale 33%
Dynarex Denture Adhesive cream is a long-lasting denture adhesive that securely holds dentures in place with a great tasting fresh minty flavor.Den...
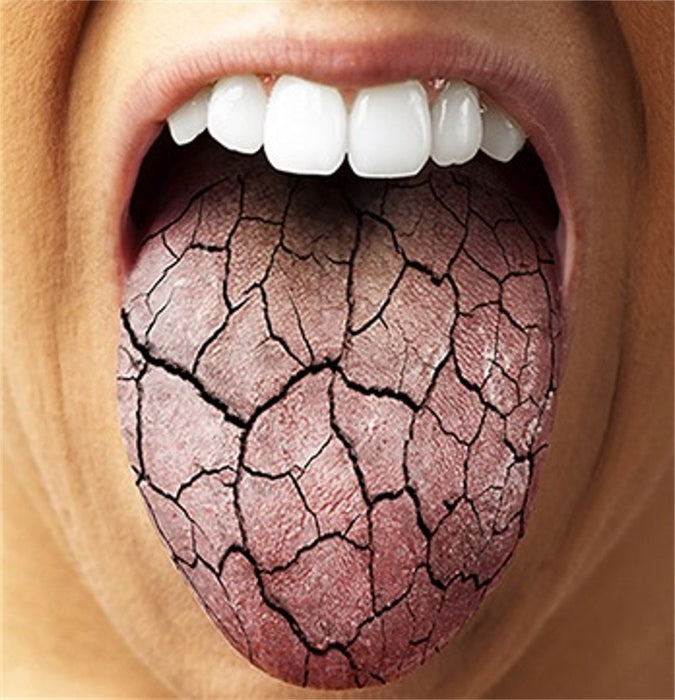
View full detailsColgate Prevident 5000 Dry Mouth is a clinically proven prescription strength toothpaste for dry mouth (xerostomia). Contains 1.1% (w/w) Sodium Fluoride has a soothing mint flavor and a unique liquid gel formula that works faster and better than other paste-form Rx toothpastes. It only needs to be used once daily and it remineralizes root caries in as little as just 3-6 months! Plus, it’s safe to use by children and adults over 6 years of age. Self-topical neutral fluoride toothpaste containing 1.1% (w/w) sodium fluoride for use as a dental caries preventive in adults and pediatric patients.
Indications and Usage:
A dental caries preventive; for once-daily self-applied topical use. PreviDent® 5000 Dry Mouth (Rx) brand of 1.1% sodium fluoride toothpaste in a squeeze bottle is easily applied onto a toothbrush. This prescription toothpaste should be used once daily in place of your regular toothpaste unless otherwise instructed by your dental professional. May be used in areas where drinking water is fluoridated since topical fluoride cannot produce fluorosis.
Contraindications:
Do not use in pediatric patients under age 6 years unless recommended by a dentist or physician.
Warnings
Prolonged daily ingestion may result in various degrees of dental fluorosis in pediatric patients under age 6 years, especially if the water fluoridation exceeds 0.6 ppm, since younger pediatric patients frequently cannot perform the brushing process without significant swallowing. Use in pediatric patients under age 6 years requires special supervision to prevent repeated swallowing of toothpaste which could cause dental fluorosis. Pediatric patients under age 12 should be supervised in the use of this product. Read directions carefully before using. Keep out of reach of infants and children.
Precautions
General: Not for systemic treatment. DO NOT SWALLOW.
Carcinogenesis, Mutagenesis, Impairment of Fertility: In a study conducted in rodents, no carcinogenesis was found in male and female mice and female rats treated with fluoride at dose levels ranging from 4.1 to 9.1 mg/kg of body weight. Equivocal evidence of carcinogenesis was reported in male rats treated with 2.5 and 4.1 mg/kg of body weight. In a second study, no carcinogenesis was observed in rats, males or females, treated with fluoride up to 11.3 mg/kg of body weight. Epidemiological data provide no credible evidence for an association between fluoride, either naturally occurring or added to drinking water, and risk of human cancer.
Fluoride ion is not mutagenic in standard bacterial systems. It has been shown that fluoride ion has potential to induce chromosome aberrations in cultured human and rodent cells at doses much higher than those to which humans are exposed. In vivo data are conflicting. Some studies report chromosome damage in rodents, while other studies using similar protocols report negative results.
Potential adverse reproductive effects of fluoride exposure in humans has not been adequately evaluated. Adverse effects on reproduction were reported for rats, mice, fox, and cattle exposed to 100 ppm or greater concentrations of fluoride in their diet or drinking water. Other studies conducted in rats demonstrated that lower concentrations of fluoride (5 mg/kg of body weight) did not result in impaired fertility and reproductive capabilities.
Pregnancy - Teratogenic Effects: Pregnancy Category B. It has been shown that fluoride crosses the placenta of rats, but only 0.01% of the amount administered is incorporated in fetal tissue. Animal studies (rats, mice, rabbits) have shown that fluoride is not a teratogen. Maternal exposure to 12.2 mg fluoride/kg of body weight (rats) or 13.1 mg/kg of body weight (rabbits) did not affect the litter size or fetal weight and did not increase the frequency of skeletal or visceral malformations. There are no adequate and well-controlled studies in pregnant women. However, epidemiological studies conducted in areas with high levels of naturally fluoridated water showed no increase in birth defects. Heavy exposure to fluoride during in utero development may result in skeletal fluorosis which becomes evident in childhood.
Nursing Mothers: It is not known if fluoride is excreted in human milk. However, many drugs are excreted in milk, and caution should be exercised when products containing fluoride are administered to a nursing woman. Reduced milk production was reported in farm-raised fox when the animals were fed a diet containing a high concentration of fluoride (98-137 mg/kg of body weight). No adverse effects on parturition, lactation, or offspring were seen in rats administered fluoride up to 5 mg/kg of body weight.
Pediatric Use: The use of PreviDent® 5000 Dry Mouth (Rx) in pediatric age groups 6 to 16 years as a caries preventive is supported by pioneering clinical studies with 1.1% sodium fluoride gels in mouth trays in students age 11 to 14 years conducted by Englander et al.6-8 Safety and effectiveness in pediatric patients below the age of 6 years have not been established. Please refer to the CONTRAINDICATIONS and WARNINGS sections.
Geriatric Use: Of the total number of subjects in clinical studies of 1.1% (w/v) sodium fluoride, 15 percent were 65 and over, while 1 percent were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.9
Adverse Reactions
Allergic reactions and other idiosyncrasies have been rarely reported.
Overdosage
Accidental ingestion of large amounts of fluoride may result in acute burning in the mouth and sore tongue. Nausea, vomiting, and diarrhea may occur soon after ingestion (within 30 minutes) and are accompanied by salivation, hematemesis, and epigastric cramping abdominal pain. These symptoms may persist for 24 hours. If less than 5 mg fluoride/kg body weight (i.e., less than 2.3 mg fluoride/lb body weight) have been ingested, give calcium (e.g., milk) orally to relieve gastrointestinal symptoms and observe for a few hours. If more than 5 mg fluoride/kg body weight (i.e., more than 2.3 mg fluoride/lb body weight) have been ingested, induce vomiting, give orally soluble calcium (e.g., milk, 5% calcium gluconate or calcium lactate solution) and immediately seek medical assistance. For accidental ingestion of more than 15 mg fluoride/kg of body weight (i.e., more than 6.9 mg fluoride/lb body weight), induce vomiting and admit immediately to a hospital facility.
Administration and Dosage Instructions
Follow these instructions unless otherwise instructed by your dental professional:
Adults and pediatric patients 6 years of age or older, apply a thin ribbon of PreviDent® 5000 Dry Mouth (Rx) to a toothbrush. Brush thoroughly once daily for two minutes, preferably at bedtime, in place of your regular toothpaste.
After use, adults expectorate. For best results, do not eat, drink, or rinse for 30 minutes. Pediatric patients, ages 6-16 years, expectorate after use and rinse mouth thoroughly.
Active Ingredient – Sodium fluoride 1.1% (w/w)
Inactive Ingredients – Water, sorbitol, hydrated silica, propylene glycol, glycerin, PEG-40 hydrogenated castor oil, dipotassium phosphate, poloxamer 407, flavor, PVM/MA copolymer, xanthan gum, sodium benzoate, sodium hydroxide, sodium saccharin, cocamidopropyl betaine, cetylpyridinium chloride, potassium sorbate, pectin, FD&C Blue No. 1
Manufacturer: Colgate Oral Pharmaceutical
Size: 100 mL
No matter what is causing your dry mouth symptoms, this prescription strength Colgate Prevident 5000 Dry Mouth Gel is an easy and effective at-home self treatment for combating the effects of dry mouth. Order yours today by placing an order using our conveniently easy to use website or calling us at 1-888-687-4334 to speak with one of our friendly and experienced medical supply specialists and place an order by phone.
Buy Colgate Prevident 5000 Dry Mouth Toothpaste Gel online at Mountainside Medical Equipment.
Colgate Prevident 5000 Dry Mouth is a clinically proven prescription strength toothpaste for dry mouth (xerostomia). Contains 1.1% (w/w) Sodium Fluoride has a soothing mint flavor and a unique liquid gel formula that works faster and better than other paste-form Rx toothpastes. It only needs to be used once daily and it remineralizes root caries in as little as just 3-6 months! Plus, it’s safe to use by children and adults over 6 years of age. Self-topical neutral fluoride toothpaste containing 1.1% (w/w) sodium fluoride for use as a dental caries preventive in adults and pediatric patients.
Indications and Usage:
A dental caries preventive; for once-daily self-applied topical use. PreviDent® 5000 Dry Mouth (Rx) brand of 1.1% sodium fluoride toothpaste in a squeeze bottle is easily applied onto a toothbrush. This prescription toothpaste should be used once daily in place of your regular toothpaste unless otherwise instructed by your dental professional. May be used in areas where drinking water is fluoridated since topical fluoride cannot produce fluorosis.
Contraindications:
Do not use in pediatric patients under age 6 years unless recommended by a dentist or physician.
Warnings
Prolonged daily ingestion may result in various degrees of dental fluorosis in pediatric patients under age 6 years, especially if the water fluoridation exceeds 0.6 ppm, since younger pediatric patients frequently cannot perform the brushing process without significant swallowing. Use in pediatric patients under age 6 years requires special supervision to prevent repeated swallowing of toothpaste which could cause dental fluorosis. Pediatric patients under age 12 should be supervised in the use of this product. Read directions carefully before using. Keep out of reach of infants and children.
Precautions
General: Not for systemic treatment. DO NOT SWALLOW.
Carcinogenesis, Mutagenesis, Impairment of Fertility: In a study conducted in rodents, no carcinogenesis was found in male and female mice and female rats treated with fluoride at dose levels ranging from 4.1 to 9.1 mg/kg of body weight. Equivocal evidence of carcinogenesis was reported in male rats treated with 2.5 and 4.1 mg/kg of body weight. In a second study, no carcinogenesis was observed in rats, males or females, treated with fluoride up to 11.3 mg/kg of body weight. Epidemiological data provide no credible evidence for an association between fluoride, either naturally occurring or added to drinking water, and risk of human cancer.
Fluoride ion is not mutagenic in standard bacterial systems. It has been shown that fluoride ion has potential to induce chromosome aberrations in cultured human and rodent cells at doses much higher than those to which humans are exposed. In vivo data are conflicting. Some studies report chromosome damage in rodents, while other studies using similar protocols report negative results.
Potential adverse reproductive effects of fluoride exposure in humans has not been adequately evaluated. Adverse effects on reproduction were reported for rats, mice, fox, and cattle exposed to 100 ppm or greater concentrations of fluoride in their diet or drinking water. Other studies conducted in rats demonstrated that lower concentrations of fluoride (5 mg/kg of body weight) did not result in impaired fertility and reproductive capabilities.
Pregnancy - Teratogenic Effects: Pregnancy Category B. It has been shown that fluoride crosses the placenta of rats, but only 0.01% of the amount administered is incorporated in fetal tissue. Animal studies (rats, mice, rabbits) have shown that fluoride is not a teratogen. Maternal exposure to 12.2 mg fluoride/kg of body weight (rats) or 13.1 mg/kg of body weight (rabbits) did not affect the litter size or fetal weight and did not increase the frequency of skeletal or visceral malformations. There are no adequate and well-controlled studies in pregnant women. However, epidemiological studies conducted in areas with high levels of naturally fluoridated water showed no increase in birth defects. Heavy exposure to fluoride during in utero development may result in skeletal fluorosis which becomes evident in childhood.
Nursing Mothers: It is not known if fluoride is excreted in human milk. However, many drugs are excreted in milk, and caution should be exercised when products containing fluoride are administered to a nursing woman. Reduced milk production was reported in farm-raised fox when the animals were fed a diet containing a high concentration of fluoride (98-137 mg/kg of body weight). No adverse effects on parturition, lactation, or offspring were seen in rats administered fluoride up to 5 mg/kg of body weight.
Pediatric Use: The use of PreviDent® 5000 Dry Mouth (Rx) in pediatric age groups 6 to 16 years as a caries preventive is supported by pioneering clinical studies with 1.1% sodium fluoride gels in mouth trays in students age 11 to 14 years conducted by Englander et al.6-8 Safety and effectiveness in pediatric patients below the age of 6 years have not been established. Please refer to the CONTRAINDICATIONS and WARNINGS sections.
Geriatric Use: Of the total number of subjects in clinical studies of 1.1% (w/v) sodium fluoride, 15 percent were 65 and over, while 1 percent were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.9
Adverse Reactions
Allergic reactions and other idiosyncrasies have been rarely reported.
Overdosage
Accidental ingestion of large amounts of fluoride may result in acute burning in the mouth and sore tongue. Nausea, vomiting, and diarrhea may occur soon after ingestion (within 30 minutes) and are accompanied by salivation, hematemesis, and epigastric cramping abdominal pain. These symptoms may persist for 24 hours. If less than 5 mg fluoride/kg body weight (i.e., less than 2.3 mg fluoride/lb body weight) have been ingested, give calcium (e.g., milk) orally to relieve gastrointestinal symptoms and observe for a few hours. If more than 5 mg fluoride/kg body weight (i.e., more than 2.3 mg fluoride/lb body weight) have been ingested, induce vomiting, give orally soluble calcium (e.g., milk, 5% calcium gluconate or calcium lactate solution) and immediately seek medical assistance. For accidental ingestion of more than 15 mg fluoride/kg of body weight (i.e., more than 6.9 mg fluoride/lb body weight), induce vomiting and admit immediately to a hospital facility.
Administration and Dosage Instructions
Follow these instructions unless otherwise instructed by your dental professional:
Adults and pediatric patients 6 years of age or older, apply a thin ribbon of PreviDent® 5000 Dry Mouth (Rx) to a toothbrush. Brush thoroughly once daily for two minutes, preferably at bedtime, in place of your regular toothpaste.
After use, adults expectorate. For best results, do not eat, drink, or rinse for 30 minutes. Pediatric patients, ages 6-16 years, expectorate after use and rinse mouth thoroughly.
Active Ingredient – Sodium fluoride 1.1% (w/w)
Inactive Ingredients – Water, sorbitol, hydrated silica, propylene glycol, glycerin, PEG-40 hydrogenated castor oil, dipotassium phosphate, poloxamer 407, flavor, PVM/MA copolymer, xanthan gum, sodium benzoate, sodium hydroxide, sodium saccharin, cocamidopropyl betaine, cetylpyridinium chloride, potassium sorbate, pectin, FD&C Blue No. 1
Manufacturer: Colgate Oral Pharmaceutical
Size: 100 mL
No matter what is causing your dry mouth symptoms, this prescription strength Colgate Prevident 5000 Dry Mouth Gel is an easy and effective at-home self treatment for combating the effects of dry mouth. Order yours today by placing an order using our conveniently easy to use website or calling us at 1-888-687-4334 to speak with one of our friendly and experienced medical supply specialists and place an order by phone.
Buy Colgate Prevident 5000 Dry Mouth Toothpaste Gel online at Mountainside Medical Equipment.
Something went wrong. Please try again.
Check your email for exclusive offers and updates from Mountainside Medical.
