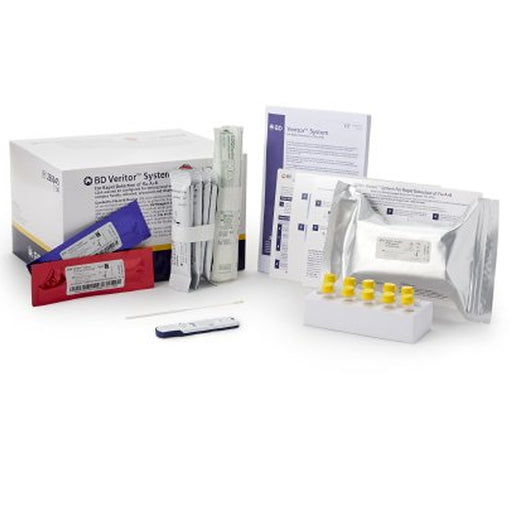
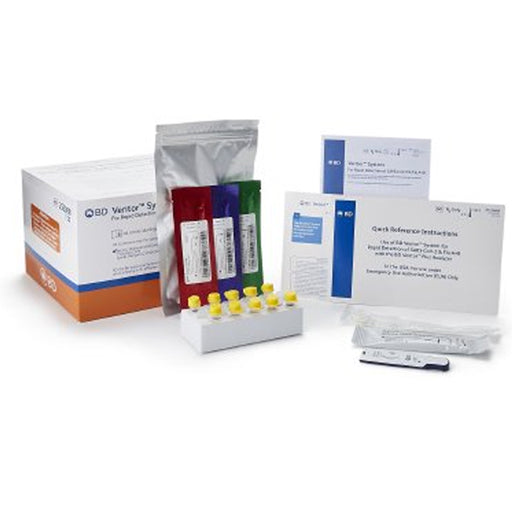
The Consult COVID-19 & Flu A&B Antigen Respiratory Test Kit (Professional Use) is a rapid point-of-care diagnostic kit designed to quickly detect and differentiate between COVID-19 (SARS-CoV-2), Influenza A, and Influenza B infections using upper respiratory (nasal) specimens. This kit is intended for use by healthcare professionals in clinical or near-patient settings, to assist in the diagnosis of active respiratory infections with similar symptoms.
Purpose
- CLIA Waived: This means the test is simple and has a low risk for an incorrect result, so it can be performed in labs or facilities with a CLIA waiver (such as doctors’ offices, clinics, or urgent care centers).
- Rapid Antigen Detection: Provides results within about 10-20 minutes for all three viruses by detecting their specific antigens in patient nasal swab samples.
Kit Contents (for 25 tests):
-
25 Sealed Test Cassettes
- Each is a self-contained device (similar to a pregnancy test) that displays the test result visually.
-
25 Sterile Nasal Swabs
- For collecting patient specimens from the anterior nose.
-
25 Pre-Filled Extraction Tubes
- Tubes already filled with buffer solution, used to mix and extract the sample from the swab.
-
25 Extraction Tube Tips
- Caps that allow you to apply drops from the tube onto the test cassette.
-
2 Tube Holders
- Racks or stands to hold tubes upright during the test process.
-
1 Quick Reference Instructions
- Laminated or summarized sheet with step-by-step images or bullets for easy use at testing stations.
-
1 Instructions for Use (IFU)
- The full operating manual, including limitations, storage, and troubleshooting information.
How It Works (General Steps)
-
Collect Sample:
- Use a sterile nasal swab to collect a sample from both nostrils.
-
Extract Sample:
- Insert swab into a pre-filled extraction tube, swirl/mix, then remove and dispose of the swab.
-
Apply to Cassette:
- Attach the tube tip to the tube and apply a few drops to the test cassette.
-
Wait for Results:
- Wait 10–20 minutes; lines appear indicating if the specimen is positive/negative for COVID-19, Influenza A, or Influenza B.
-
Interpret Results:
- Use the visual guidance (lines on the cassette) and refer to instructions to determine results.
Intended Users & Regulatory Note
- For Professional Use Only: Not intended for use at home or by untrained personnel.
- CLIA-waived: Suitable for waived laboratories/clinics with minimal regulatory requirements.
- Always refer to each kit’s FDA Emergency Use Authorization (EUA) and follow all applicable local regulations for reporting and use.
This test kit is a convenient, all-in-one package for rapidly screening patients for COVID-19, Influenza A, and Influenza B in clinics, urgent care, or doctor’s offices—and is meant to help direct further care, isolation, or confirmatory testing as needed.