Sterile Glass Vials (Medical Grade)
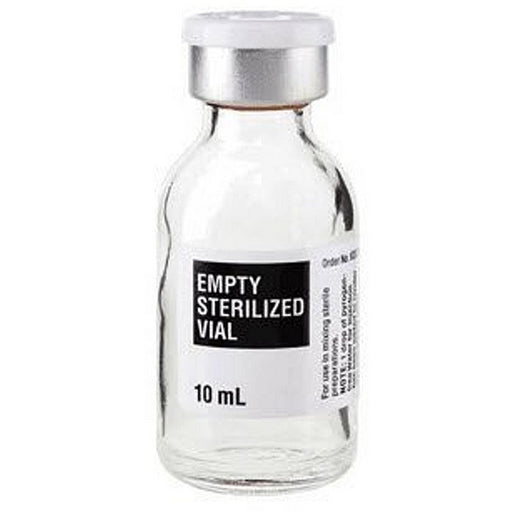
Sterile empty 10 mL glass vials are pharmaceutical containers made of medical-grade glass, used for storing injectable medications, vaccines, and sterile solutions. These single-use vials are Type I borosilicate glass, which is highly inert and chemically resistant. Borosilicate (Type I) glass contains boric oxide and alumina and offers high hydrolytic resistance, making it suitable for parenteral (injectable) products. In fact, pharmacopeias specify that Type I glass vials are generally used to package preparations intended for parenteral administration. The glass is typically clear (colorless) or sometimes amber (for light-sensitive drugs). The inside surface is usually untreated (truly “neutral” glass) so it does not interact with the vial’s content.
Sterile Empty Glass Vial for Injection that meets CGMP Requirement Standards. Empty Injection Vials are sterile and nonpyrogenic. Used for mixing different medications and drugs for injection. The vials contain a plastic protective cap which can be removed prior to using the vials. The sealed top provides easy needle insertion, while keeping the injected liquids safely contained for mixing.
- cGMP Compliant
- Vial Size 20mm x 52mm
- Drug Master File (DMF) III Compliant
Benefits:
- Pyrogen free
- Aluminum sealed top
- Clear glass
- Butyl compound (synthetic rubber) stopper
- Blue flip-top cap
Each 10 mL vial is precisely engineered for sterile use. A typical vial has a nominal volume of 10 mL but a “brimful” volume of about 13.5 mL (the amount the vial actually holds to the brim). The outer diameter is around 24 mm and height about 45 mm for a standard 20 mm-closure vial. (Indeed, 10 mL vials commonly use a 20 mm neck finish.) All vials are manufactured to pharmaceutical cleanroom standards (GMP) and are pyrogen-tested and certified sterile. In practice, manufacturers wash the vials with Water-for-Injection (WFI), dry them with sterile filtered air, and then sterilize them (typically by autoclaving or gamma irradiation) before final packaging. Vendor certificates often accompany each lot, confirming sterility and absence of pyrogens (fever-causing bacterial endotoxins).
Construction: The vial itself is closed with a rubber stopper (usually a butyl rubber) and an aluminum cap. The rubber stopper provides a resealable, needle‐pierceable seal, and the aluminum crimp (often with a colored plastic flip-off top) secures the stopper in place. For example, Thermo Scientific’s sterile vials are explicitly described as “Type I borosilicate, assembled with butyl stoppers and aluminum seals”. This combination is standard: it maintains an airtight, sterile barrier while allowing syringe access. The vials are non-reactive with water or aqueous drugs, so contents remain uncontaminated and stable.
Uses and Applications
Sterile 10 mL glass vials are used wherever a small, sealed container is needed for injectable substances or other sterile fluids. Common uses include:
- Drug reconstitution/mixing: Many injectable medications are supplied as dry powders. A pharmacist or clinician adds diluent (e.g. sterile saline) into a sterile vial to dissolve the drug.
- Parenteral drug dispensing: Hospitals and compounding pharmacies use these vials to dispense patient-dosed solutions (after aseptic preparation) for injection or infusion.
- Vaccine and biologics packaging: Vaccines, monoclonal antibodies, and other biotherapeutics are often filled into sterile vials in vial fill-finish lines.
- Diagnostic reagents: Clinical labs may use sterile vials for blood or urine standards, reference reagents, or sample storage (when sterility is required).
- Research and specialty products: Laboratories use sterile vials for cell culture additives, enzyme preparations, or any fluid that must remain sterile and uncontaminated.
In all cases, the vial’s seal keeps contents sterile until use. Users remove the plastic flip-off cap (if present) and insert a sterile needle through the rubber stopper to aspirate or inject the fluid. Because the glass and stopper are pyrogen-free, they will not introduce toxins or particulates into the solution.
Specifications and Standards
Key specifications for medical 10 mL vials include:
- Type I borosilicate glass: As noted, this is required for injectable use due to its chemical inertness and proven compatibility with parenteral drugs. Type II (treated soda-lime) is only used in special cases, and Type III (ordinary soda-lime) is generally not acceptable for injectables.
- Pyrogen-free and sterile: By pharmacopeial standards, vials must be depyrogenated (no endotoxin) and terminally sterilized. Suppliers provide sterility certificates for each batch.
- Closure size: For 10 mL vials, the standard finishing is a 20 mm diameter neck. This matches commonly used cap and stoppering equipment.
- Volume markings: Vials may have gradations or simply a nominal volume. A 10 mL vial typically holds about 13–14 mL to the brim.
- Packaging: Sterile vials are usually nested in trays or “tubs” covered with Tyvek™ or film, and shipped in sterile barrier packaging. This packaging maintains sterility until opened in the clean setting.
Because these vials are a primary container for sterile products, manufacturing and quality control follow strict regulations (e.g. ISO 15378 for primary packaging, cGMP for aseptic fill). Many vendors list a Drug Master File (DMF) number for their sterile vials, underscoring compliance with health authorities.
Care and Handling
Empty sterile vials must be handled aseptically. Before filling, an operator checks for flaws (no cracks, chips, or particulate matter inside). The stopper and cap should remain sealed until ready for use to keep the contents sterile. Once the seal is breached (needle insertion), the contents should be used promptly according to sterile compounding guidelines. Discard any unused portion unless validated for storage, as contamination can occur after opening.
A single 10 mL vial is intended for one filling. If only part of the vial is used, any remaining solution must be re-tested or discarded, depending on the drug’s stability. Any unused empty vials should be sanitized or disposed of per facility protocols.
Summary
In summary, a sterile 10 mL glass vial is a small Type I borosilicate container with a butyl rubber stopper and aluminum crimp cap. It meets pharmaceutical standards for sterility and pyrogen removal. These vials are widely used for injectable drugs, biologics, and clinical samples. Their inert glass and quality closure system ensure that medications remain safe and uncontaminated until the moment of injection. Always follow clinical guidelines for sterile technique when using these vials.