Clonidine 0.1 mg Tablets 100/Bottle (RX)
How to Order:
You will receive instructions on how to create an account along with Rx Ordering Details.
(Note: Acceptable licenses must have Prescriptive Authority in the license issuing state.)
Cart subtotal





 Sale 33%
Sale 33%
Powerful Oral Solution for Hair Loss, Androgenetic Alopecia, and Resistant Hypertension Unlock the next level of hair regrowth and blood pressure m...
View full details Sale 58%
Sale 58%
Adenosine Injection is a medication form of adenosine that is used primarily in emergency and hospital settings to treat certain types of abnormal ...
View full details Sale 48%
Sale 48%
Nitroglycerin Sublingual Tablets 0.3 mg are prescribed for the acute relief of an angina pectoris attack, characterized by chest pain when the hear...
View full details Sale 42%
Sale 42%
Labetalol Hydrochloride (HCL) injection, at a concentration of 5 mg/mL, is a medication formulated to provide effective management of acute hyperte...
View full details Sale 38%
Sale 38%
Comprehensive Cardiac, Blood Pressure & Migraine Protection – Trusted Prescription-Strength Beta Blocker Take charge of your heart health, bloo...
View full details Sale 22%
Sale 22%
Atenolol Tablets 50 mg (by Aurobindo Pharma) are a prescription medication used to treat high blood pressure, chest pain (angina), and other heart ...
View full details Sale 22%
Sale 22%
Atenolol Tablets 25 mg (by Aurobindo Pharma) are a prescription medication used to treat high blood pressure, chest pain (angina), and other heart ...
View full details Sale 50%
Sale 50%
Minoxidil Tablets 2.5 mg by Actavis offer advanced, prescription-only treatment for severe or resistant hypertension (high blood pressure) when sta...
View full details Sale 32%
Sale 32%
#1 Prescription Blood Pressure Medication — Effective Hypertension Control & Kidney Protection with Irbesartan 150 mg Take charge of your heart...
View full details Sale 31%
Sale 31%
Omega-3-Acid Ethyl Esters Capsules 1 gram is a prescription medication primarily used to lower high triglyceride levels in adults. It contains esse...
View full details Sale 33%
Sale 33%
Powerful Oral Solution for Hair Loss, Androgenetic Alopecia, and Resistant Hypertension Unlock the next level of hair regrowth and blood pressure m...
View full details Sale 48%
Sale 48%
Nitroglycerin Sublingual Tablets 0.3 mg are prescribed for the acute relief of an angina pectoris attack, characterized by chest pain when the hear...
View full details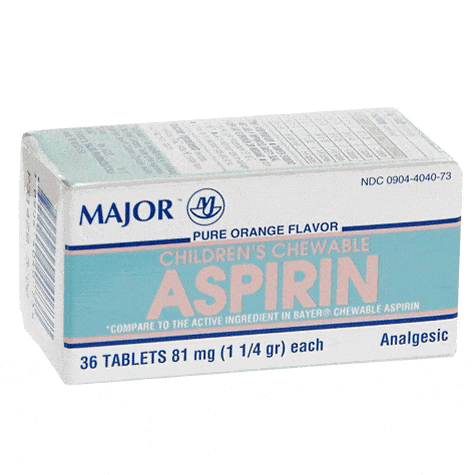 Sale
Sale
Chewable Low Dose Baby Aspirin 81mg are an effective pain reliever for children. This pain relief medication features a kid-friendly chewable consi...
View full details Sale 42%
Sale 42%
Labetalol Hydrochloride (HCL) injection, at a concentration of 5 mg/mL, is a medication formulated to provide effective management of acute hyperte...
View full details Sale 33%
Sale 33%
Metoprolol Tartrate for Injection is used primarily to manage acute episodes of hypertension and to control heart rate in patients experiencing tac...
View full details Sale 38%
Sale 38%
Comprehensive Cardiac, Blood Pressure & Migraine Protection – Trusted Prescription-Strength Beta Blocker Take charge of your heart health, bloo...
View full details Sale 25%
Sale 25%
Vitamin C 500 mg with Rose Hips Chewable Tablets, Orange Flavor is a dietary supplement that provides 500 mg of vitamin C per tablet. It also conta...
View full details Sale 22%
Sale 22%
Atenolol Tablets 50 mg (by Aurobindo Pharma) are a prescription medication used to treat high blood pressure, chest pain (angina), and other heart ...
View full details Sale 38%
Sale 38%
Heparin Sodium Injection (40,000 Units) Heparin sodium injection is a powerful anticoagulant (blood thinner) given by a healthcare professional. It...
View full details Sale 22%
Sale 22%
Atenolol Tablets 25 mg (by Aurobindo Pharma) are a prescription medication used to treat high blood pressure, chest pain (angina), and other heart ...
View full details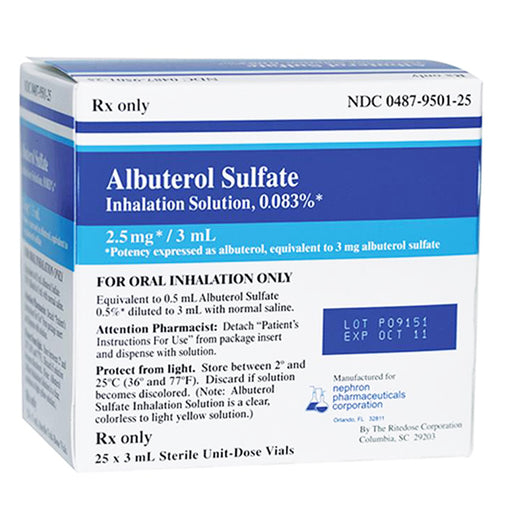 Sale 38%
Sale 38%
Fast-Acting Relief for Asthma, COPD, and Bronchospasm – Professional-Grade Albuterol Nebulizer Solution Breathe easier and faster with Albuterol Su...
View full details Sale 20%
Sale 20%
Hylenex Injection Hylenex is the brand name for a recombinant human hyaluronidase enzyme given by injection. Hyaluronidase is sometimes nicknamed a...
View full details Sale 24%
Sale 24%
Naloxone prefilled syringe refers to a ready-to-use syringe that comes pre-loaded with naloxone solution, intended for quick administration during ...
View full details Sale 23%
Sale 23%
Lidocaine 1% Injection Lidocaine (also called lignocaine) 1% injection is a local anesthetic solution (10 mg of lidocaine per mL) used to numb tiss...
View full details Sold out
Sold out
Kenalog 40 Injection (40 mg per 1 mL) is a corticosteroid used to treat various conditions involving inflammation and allergic reactions, such as a...
View full details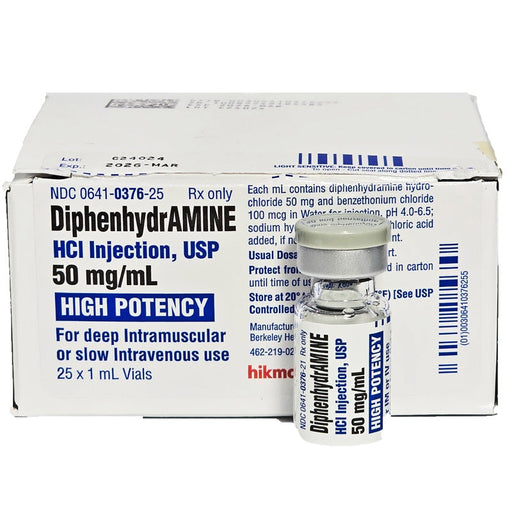 Sale
Sale
Diphenhydramine 50 mg Diphenhydramine Injection 50 mg is a powerful, fast-acting solution for acute allergy relief. As an injectable form of the fa...
View full details Sale 41%
Sale 41%
Fast-Acting Local Anesthetic for Pain Management & Medical Procedures Lidocaine Hydrochloride Injection 1% (10 mg/mL), 30 mL Single-Dose Vial b...
View full details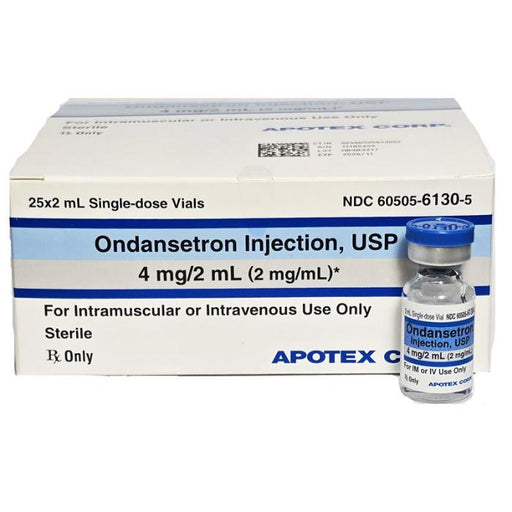 Sale 46%
Sale 46%
Ondansetron Injection is used to prevent nausea and vomiting associated with chemotherapy, radiation therapy, and surgery. It works by blocking ser...
View full details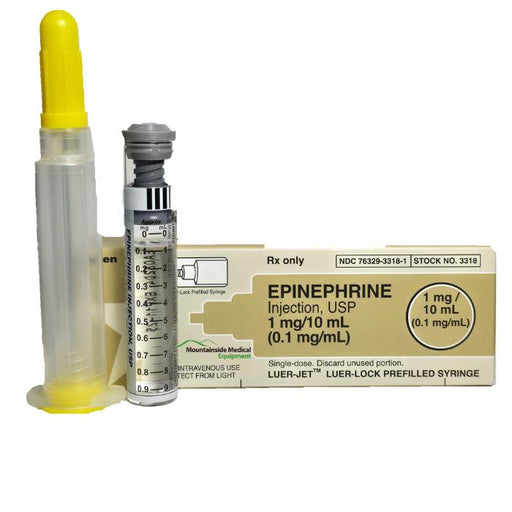 Sale 31%
Sale 31%
An Epinephrine Prefilled Syringe is a medical device containing a specific dose of epinephrine (also known as adrenaline), already loaded into a sy...
View full details Sale 7%
Sale 7%
Ondansetron is a medication primarily used to prevent nausea and vomiting caused by cancer chemotherapy, radiation therapy, or surgery. Ondansetron...
View full detailsClonidine is a centrally acting α₂-adrenergic (and imidazoline) receptor agonist. It lowers blood pressure by decreasing sympathetic outflow from the brainstem, causing vasodilation and a slower heart rate. In other words, clonidine stimulates α₂ receptors in the central nervous system, which reduces norepinephrine release – allowing blood vessels to relax and the heart to beat more slowly.
Clonidine is FDA-approved primarily as an antihypertensive agent. It is typically used when blood pressure is not adequately controlled with first-line drugs (a “last-line” add-on for resistant hypertension). Other approved and common uses include:
Hypertension: As monotherapy or (more often) as an add-on drug to control high blood pressure. It relaxes both arteries and heart muscle to reduce BP and pulse.
ADHD (children): An extended-release form (Kapvay) is approved for pediatric attention-deficit/hyperactivity disorder. Kapvay helps reduce hyperactivity/impulsivity when stimulants are insufficient.
Tourette’s syndrome: Adjunct therapy to help control tics in patients with Tourette’s (clonidine is indicated for tics).
Severe pain (cancer): Off-label as an analgesic adjuvant – clonidine may be used in anesthesia or cancer pain to potentiate pain control (its sedative/analgesic properties can help in refractory pain situations).
Withdrawal syndromes (off-label): Clonidine is commonly used to alleviate withdrawal symptoms from opioids, alcohol, or benzodiazepines. (For example, it’s used in detox and neonatal withdrawal protocols due to its ability to blunt sympathetic “crash” symptoms.)
Other off-label uses: Menopausal hot flashes, migraine prophylaxis, restless legs, etc., have been described in the literature. (Some sources list acute hypertension, pheochromocytoma crisis, erectile dysfunction, and others under investigational/limited use.
Dosage must be individualized and titrated carefully. Typical dosing guidelines are:
Adults (immediate-release tablets): Start 0.1 mg orally twice daily (once in the morning and once at bedtime). Because clonidine can cause drowsiness, many prescribers give the larger portion of the dose at bedtime. Elderly or frail patients often start at 0.1 mg once daily.
Titration: Increase in 0.1 mg increments per week as needed to reach target BP. The maintenance dose is usually 0.2–0.6 mg/day given in divided doses. (For example, a schedule might end up 0.2 mg in AM and 0.4 mg in PM, etc.) Adjust or split doses according to response. Do not exceed the prescribed maximum.
Extended-release (Kapvay) for ADHD: Common starting dose is 0.1 mg at bedtime (especially in children), which can be increased by 0.1 mg every 3–7 days if needed. Total daily doses above 0.4 mg/day (0.2 mg twice daily) are generally not used for ADHD. Always swallow ER tablets whole (do not crush/chew).
Renal/hepatic impairment: May require lower doses or slower titration.
As patch: (for reference) Clonidine is also available as a transdermal patch (0.1 mg/24 hr and up). If switching from oral to patch, follow specific product guidelines. Do not use an oral tablet and a patch simultaneously (risk of overdose).
Monitoring: BP and heart rate should be checked frequently during dose adjustments. Follow doctor’s instructions exactly.
Clonidine’s side effects largely reflect its CNS and cardiovascular actions. Frequent adverse effects include:
Central nervous system: Drowsiness, dizziness, sedation and fatigue (often most pronounced during initiation or dose increases). Insomnia or vivid dreams can occur paradoxically, but overall CNS depression is common. (Patients should be cautioned about driving or operating machinery until they see how the drug affects them.
Dry mouth: Very common. Keep hydrated and use sugarless gum or lozenges as needed.
Gastrointestinal: Constipation, decreased appetite/nausea (loss of appetite).
Cardiovascular: Bradycardia (slow heart rate) and hypotension can occur. Patients may feel lightheaded or faint, especially on standing.
Others: Possible rash, erectile dysfunction, irritability or mood changes, muscle weakness, weight gain, headache. (Rarely depression may worsen or appear with long-term use.)
Rebound effects: If clonidine is missed or abruptly stopped, acute rebound hypertension with nervousness and headache may occur. Slow tapering is essential.
Key Points: Always follow prescription instructions and never self-medicate or adjust dose without medical guidance. Because of its significant side effects (especially sedation) and rebound risk, clonidine therapy should be managed by a healthcare professional. Patients should be informed about avoiding sudden discontinuation, limiting alcohol, and not engaging in hazardous activities until they know how clonidine affects them.
Clonidine is a centrally acting α₂-adrenergic (and imidazoline) receptor agonist. It lowers blood pressure by decreasing sympathetic outflow from the brainstem, causing vasodilation and a slower heart rate. In other words, clonidine stimulates α₂ receptors in the central nervous system, which reduces norepinephrine release – allowing blood vessels to relax and the heart to beat more slowly.
Clonidine is FDA-approved primarily as an antihypertensive agent. It is typically used when blood pressure is not adequately controlled with first-line drugs (a “last-line” add-on for resistant hypertension). Other approved and common uses include:
Hypertension: As monotherapy or (more often) as an add-on drug to control high blood pressure. It relaxes both arteries and heart muscle to reduce BP and pulse.
ADHD (children): An extended-release form (Kapvay) is approved for pediatric attention-deficit/hyperactivity disorder. Kapvay helps reduce hyperactivity/impulsivity when stimulants are insufficient.
Tourette’s syndrome: Adjunct therapy to help control tics in patients with Tourette’s (clonidine is indicated for tics).
Severe pain (cancer): Off-label as an analgesic adjuvant – clonidine may be used in anesthesia or cancer pain to potentiate pain control (its sedative/analgesic properties can help in refractory pain situations).
Withdrawal syndromes (off-label): Clonidine is commonly used to alleviate withdrawal symptoms from opioids, alcohol, or benzodiazepines. (For example, it’s used in detox and neonatal withdrawal protocols due to its ability to blunt sympathetic “crash” symptoms.)
Other off-label uses: Menopausal hot flashes, migraine prophylaxis, restless legs, etc., have been described in the literature. (Some sources list acute hypertension, pheochromocytoma crisis, erectile dysfunction, and others under investigational/limited use.
Dosage must be individualized and titrated carefully. Typical dosing guidelines are:
Adults (immediate-release tablets): Start 0.1 mg orally twice daily (once in the morning and once at bedtime). Because clonidine can cause drowsiness, many prescribers give the larger portion of the dose at bedtime. Elderly or frail patients often start at 0.1 mg once daily.
Titration: Increase in 0.1 mg increments per week as needed to reach target BP. The maintenance dose is usually 0.2–0.6 mg/day given in divided doses. (For example, a schedule might end up 0.2 mg in AM and 0.4 mg in PM, etc.) Adjust or split doses according to response. Do not exceed the prescribed maximum.
Extended-release (Kapvay) for ADHD: Common starting dose is 0.1 mg at bedtime (especially in children), which can be increased by 0.1 mg every 3–7 days if needed. Total daily doses above 0.4 mg/day (0.2 mg twice daily) are generally not used for ADHD. Always swallow ER tablets whole (do not crush/chew).
Renal/hepatic impairment: May require lower doses or slower titration.
As patch: (for reference) Clonidine is also available as a transdermal patch (0.1 mg/24 hr and up). If switching from oral to patch, follow specific product guidelines. Do not use an oral tablet and a patch simultaneously (risk of overdose).
Monitoring: BP and heart rate should be checked frequently during dose adjustments. Follow doctor’s instructions exactly.
Clonidine’s side effects largely reflect its CNS and cardiovascular actions. Frequent adverse effects include:
Central nervous system: Drowsiness, dizziness, sedation and fatigue (often most pronounced during initiation or dose increases). Insomnia or vivid dreams can occur paradoxically, but overall CNS depression is common. (Patients should be cautioned about driving or operating machinery until they see how the drug affects them.
Dry mouth: Very common. Keep hydrated and use sugarless gum or lozenges as needed.
Gastrointestinal: Constipation, decreased appetite/nausea (loss of appetite).
Cardiovascular: Bradycardia (slow heart rate) and hypotension can occur. Patients may feel lightheaded or faint, especially on standing.
Others: Possible rash, erectile dysfunction, irritability or mood changes, muscle weakness, weight gain, headache. (Rarely depression may worsen or appear with long-term use.)
Rebound effects: If clonidine is missed or abruptly stopped, acute rebound hypertension with nervousness and headache may occur. Slow tapering is essential.
Key Points: Always follow prescription instructions and never self-medicate or adjust dose without medical guidance. Because of its significant side effects (especially sedation) and rebound risk, clonidine therapy should be managed by a healthcare professional. Patients should be informed about avoiding sudden discontinuation, limiting alcohol, and not engaging in hazardous activities until they know how clonidine affects them.
Clonidine HCL Tablets 0.1 mg by Teva 100/Bottle (RX)
As always, excellent customer service and quality products!
Clonidine HCL Tablets 0.1 mg by Teva 100/Bottle (RX)
As always, excellent customer service and quality products!
Something went wrong. Please try again.
Check your email for exclusive offers and updates from Mountainside Medical.
