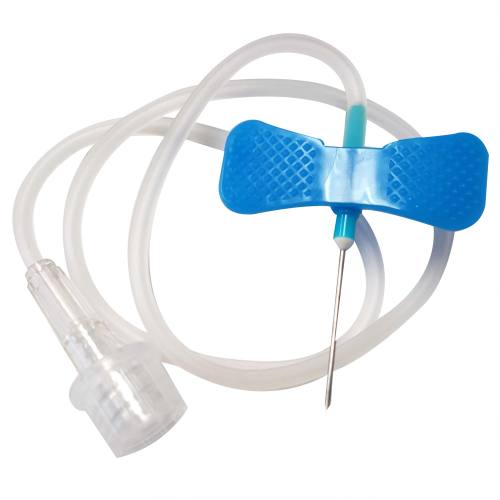
Exel IV Administration Set
Upgrade your medical supplies with the industry’s most advanced IV Administration Set, engineered for secure, accurate, and efficient intravenous therapy. Essential for hospitals, clinics, emergency medicine, and home healthcare, our IV set delivers precision, reliability, and safety for both patients and healthcare professionals.
What is an IV Administration Set?
An IV Administration Set, commonly known as an IV set or infusion set, is a medical device designed for the controlled delivery of fluids, medications, blood products, and nutrients directly into a patient’s vein. It is a critical component for parenteral therapy, resuscitation, and continuous IV infusion in diverse clinical settings.
An Intravenous Set is essential to delivering fluids and drugs directly into the bloodstream of a patient. It consists of an IV Administration Set, Y-Injection Site, 78" Tube and Roller Clamp, which work cohesively to provide these substances to the patient. The IV Administration Set is connected to an IV consulting bag and solution container, which are then suspended by an IV stand. The Y-Injection Site connects to various IV lines used to inject drugs and additives into the patient. The 78" Tube is then inserted in the patient’s vein, and the Roller Clamp, which is adjustable, is used to regulate the rate of flow. All the components of the Intravenous Set are essential for delivering fluids and drugs directly into the bloodstream of a patient.
Exel IV Administration Set Drop (micro-drip) is a clear, sterile, non-toxic iv tubing that is 78" long, used for intravenous administration. The Exel IV set is latex free has a adelberg III clamp plus an occlusion clamp that allows you to shut off the IV without having to use the roller clamp. Exel Iv sets are universal and can be attached to any IV bag quickly and easily.
Please be sure to properly select Luer Lock or Luer Slip from the drop-down box on the top right to ensure proper connection.
Features:
- Sterile
- Latex Free
- Y-injection site
- Non-pyrogenic fluid path
- Roller clamp and occlusion clamp
- Available with Luer Lock and Luer Slip connection tips. Vented and non-vented on the same set by opening and closing the valve. Exel IV Administration Sets are available in different size drops and luer connectors, and work great with any standard IV bag.
Exel IV Administration Set: Essential Infusion Equipment
The Exel IV Administration Set is a sterile, latex-free intravenous delivery system engineered for safe and efficient fluid and medication administration directly into the patient's bloodstream. This complete IV set includes all necessary components—Y-injection site, 78-inch tubing, roller clamp, and occlusion clamp—configured for universal compatibility with standard IV bags and solution containers.
This microdrip IV set (60 drops per milliliter) is designed for applications requiring precise flow control and accurate dosing. The clear, non-pyrogenic tubing allows visual monitoring of fluid flow, while the dual-clamp system provides redundant flow regulation options for clinical safety.
What Makes This IV Administration Set Complete
A functional intravenous set requires multiple integrated components working together. The IV bag or solution container hangs from an IV stand, suspending fluid above the patient. The tubing connects this container to the patient's vein, while the Y-injection site allows clinicians to inject medications and additives into the line without disrupting the primary infusion. The roller clamp regulates flow rate by compressing the tubing, and the occlusion clamp provides a secondary shut-off mechanism without relying on the roller clamp alone.
Exel's design consolidates these requirements into a single, pre-assembled unit, eliminating the need to source and connect multiple components separately. This reduces setup time and potential connection errors in clinical environments.
Key Product Features
- Sterile, non-pyrogenic tubing: Pre-sterilized and tested to eliminate fever-causing substances; safe for direct patient contact
- Latex-free construction: Eliminates risk for patients and clinicians with latex sensitivity
- 78-inch tubing length: Provides sufficient distance from IV stand to patient for mobility and positioning flexibility
- Y-injection site: Allows secondary medication administration without disconnecting primary infusion
- Roller clamp + occlusion clamp: Dual flow control mechanisms provide redundancy and precision adjustment
- Available connector options: Luer Lock (secure, threaded connection) or Luer Slip (push-fit, quick connection) for compatibility with different IV bags and devices
- Vented and non-vented capability: Single set can function in either mode by opening or closing an integrated valve
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Why Microdrip (60 Drop) IV Sets Matter
IV administration sets are classified by drop factor—the number of drops per milliliter of fluid. The Exel 60 drop set (microdrip) delivers smaller drops than macrodrip sets (10-15 drops per milliliter), enabling more precise flow control. This design is essential for medications requiring exact dosing and for patient populations sensitive to fluid volume variations.
Microdrip sets are particularly suited for medication infusions where accuracy is critical, pediatric and geriatric patients where fluid tolerance is narrow, and rapid infusion scenarios where fine control prevents overshoot.
Technical Specifications: Exel IV Administration Set
| Specification | Value | Clinical Significance |
|---|---|---|
| Drop Factor | 60 drops/mL | Microdrip configuration; enables precise flow rates (typically 1-2 mL/min increments) |
| Tubing Length | 78 inches | Sufficient clearance from IV stand to patient; allows patient repositioning and movement |
| Primary Port Type | Luer Lock or Luer Slip (selectable) | Connects to IV bag or solution container; choice affects security vs. quick-change capability |
| Y-Injection Site | Standard luer connector | Allows secondary line or push medication administration into primary infusion |
| Flow Control | Roller clamp + occlusion clamp | Dual regulation methods; occlusion clamp enables shutdown without compressing tubing |
| Tubing Material | Clear, non-pyrogenic PVC or equivalent | Visibility of fluid and air bubbles; non-pyrogenic reduces fever risk |
| Sterility | Sterile, single-use | Pre-sterilized via appropriate method (gamma radiation, ethylene oxide, or steam); must be opened and used in sterile procedure |
| Latex Content | Latex-free | Safe for all patients; eliminates hypersensitivity risk |
| Venting | Integrated valve (vented/non-vented switchable) | Vented mode: allows air in as fluid drains; non-vented: prevents backflow (use depends on IV bag type and clinical setting) |
| Connection Compatibility | Universal (standard luer connectors) | Compatible with any IV bag featuring standard luer port |
Understanding Luer Lock vs. Luer Slip
The primary connection to your IV bag is available in two configurations. Luer Lock features a threaded collar that twists onto the IV bag port, creating a secure, fixed connection that resists accidental disconnection. This configuration is preferred in critical care, transport, and high-movement scenarios where jarring or pulling on the tubing could dislodge a non-secured connection.
Luer Slip is a straight, conical fit that pushes onto the IV bag port. No threading is required, enabling faster connection and disconnection. This style is often used in rapid-infusion scenarios, emergency departments, and settings where frequent line changes occur.
Select Luer Lock if your facility prioritizes connection security and uses standard IV bags with threaded ports. Select Luer Slip if your workflow emphasizes speed of setup or if your IV containers feature slip-fit ports. Most standard IV bags are compatible with both styles; verify your bag's port type before ordering if uncertain.
Clinical Applications and Professional Use Contexts
The Exel 60-drop IV administration set is designed for healthcare settings where precision fluid delivery and medication administration are required. This section provides context for clinical decision-making regarding when this IV set is appropriate for your patient population and operational needs.
Ideal Use Scenarios
Medication Infusions: The microdrip (60 drop) design enables precise dosing for medications with narrow therapeutic windows—antibiotics, antivirals, vasopressors, insulin infusions, and chemotherapy agents. The small drop size allows clinicians to adjust flow rate in 1-2 mL/minute increments rather than larger steps, minimizing overdose or underdose risk.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Pediatric and Geriatric Populations: Patients with limited fluid tolerance benefit from microdrip precision. Pediatric patients often require exact volume calculations; elderly patients with cardiac or renal limitations tolerate narrow fluid windows. The 60-drop set prevents fluid overload by enabling precise measurement and flow control.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Rapid Infusion with Control: While not designed for emergency bolus infusion (where macrodrip 10-drop sets are preferred), the Exel set supports controlled rapid administration when higher flow is needed while maintaining drop-rate visibility and prevention of air entrainment.
Outpatient Clinics and Infusion Centers: Settings providing IV therapy outside hospitals often manage longer infusion times with lower flow rates. The Exel set's 78-inch tubing length accommodates patient positioning in recliners and allows arm mobility during extended infusions.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Latex-Sensitive Patients and Clinicians: Facilities with patients who have documented latex allergy or clinicians with latex sensitivity require latex-free equipment. The Exel IV set eliminates latex exposure throughout the infusion system.
Clinical Workflow Integration
The Y-injection site enables secondary medication administration without disconnecting the primary infusion line. This allows clinicians to deliver bolus doses, flush the line, or administer incompatible medications through the secondary port while the primary infusion continues uninterrupted. This design reduces manipulation of central lines and decreases infection risk associated with frequent connections and disconnections.
The dual-clamp system provides workflow flexibility. Most clinicians use the roller clamp for routine flow adjustment. The occlusion clamp serves as a secondary control mechanism or allows one-handed shutdown when the clinician is otherwise engaged (for example, during dressing changes or patient repositioning).
Setting-Specific Deployment
| Setting | Typical Use | Key Advantage |
|---|---|---|
| Hospital Acute Care (ICU, Medical/Surgical Floors) | Medication infusions, fluid management, multi-therapy administration via Y-site | Precision control + redundant clamp systems + rapid secondary-medication capability |
| Emergency Department | IV access, initial medication administration, fluid resuscitation (supplemented with macrodrip sets for rapid volume) | Universal compatibility + quick setup with Luer Slip option |
| Outpatient Infusion Center | Antibiotic, chemotherapy, immunotherapy, or biologic infusions; extended treatment times | Precise flow control for long-duration infusions + comfortable tubing length for patient mobility |
| Physician Office or Clinic | IV therapy, medication administration, minor procedures | Compact, self-contained unit requires no assembly + easy inventory management |
| Home Health / Visiting Nurse | Chronic infusion therapy, antibiotic or pain management infusions | Sterile, pre-assembled design reduces setup time at patient bedside + compatible with standard IV bags |
| Rehabilitation and Long-Term Care | Ongoing fluid and medication delivery in lower-acuity settings | Reliable, cost-effective solution for consistent, non-critical infusions |
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Safety Considerations and Contraindications
The Exel IV administration set is a critical medical device whose proper use directly affects patient safety. Understanding safe handling, appropriate use, and contraindications is essential before deployment in any clinical setting.
Sterility and Pre-Use Inspection
The Exel IV set is supplied sterile in sealed, single-use packaging. Do not use if the package is opened, torn, damaged, or shows signs of contamination. Before use, visually inspect the tubing for:
- Cloudiness, discolouration, or opaque sections (indicate contamination or degradation)
- Cracks, holes, or punctures in tubing (renders set unsafe for use)
- Loose connectors or damaged clamps (compromise flow control)
- Any foreign material or debris inside the tubing
If any defect is detected, discard the set and use a new, unopened package. Do not attempt to repair or reuse a damaged IV set.
Non-Pyrogenic Assurance and Fever Prevention
The Exel IV set is non-pyrogenic, meaning it has been tested to eliminate or minimise pyrogenic substances (endotoxins) that cause fever responses in patients. This is accomplished through manufacturing processes including sterile filtration, steam sterilisation, or gamma radiation. However, non-pyrogenic status does not eliminate all infection risk; aseptic technique during insertion and maintenance remains the clinician's responsibility. Contamination at the infusion site, solution container, or during secondary medication administration can still introduce pathogens or fever-causing substances.
Latex-Free Safety Profile
The Exel IV set contains no natural rubber latex. This eliminates the risk of immediate or delayed hypersensitivity reactions in latex-allergic patients and clinicians. However, confirm that all connected equipment (IV bags, secondary infusion sets, medication syringes, extension tubing) is also latex-free if the patient has documented latex allergy. A single latex-containing component anywhere in the infusion system can trigger a reaction.
Contraindications and Situations Requiring Alternative Equipment
Incompatible IV Bag Types: The Exel set requires a standard luer-port IV bag (glass or plastic container with integrated or external luer connector). It is not compatible with gravity-feed bags without ports or with non-standard connector systems. Verify connector compatibility before use.
Emergency Rapid Infusion (Massive Fluid Resuscitation): While the Exel microdrip set can deliver fluid quickly, it is not optimised for maximum-rate infusion. The 60-drop factor limits flow relative to macrodrip (10-15 drop) sets. For emergency blood transfusion, massive crystalloid resuscitation, or acute haemorrhage management, use macrodrip IV sets designed for rapid infusion. The Exel set is appropriate for controlled rapid infusion but not for uncontrolled emergency volume replacement.
Non-Vented IV Bags: The Exel set includes an integrated valve allowing vented or non-vented operation. Confirm the IV bag type before use. Rigid plastic bags require non-vented operation (air cannot enter as fluid drains); glass bottles typically require vented operation. Mismatching vented operation to bag type causes fluid flow to slow, stop, or create backflow.
Patients with Documented Connector Incompatibility: Although the set uses universal Luer connectors, rare cases of non-standard patient equipment exist. Verify all connection points before initiating infusion, particularly in patients transferred from other facilities or using home-care equipment.
Handling, Storage, and Shelf Life
Store the Exel IV set at room temperature (68–77°F / 20–25°C) in a dry environment away from direct sunlight. Protect from extreme heat, which can degrade the tubing. Most IV sets have a 3-5 year shelf life from the manufacturer's date; verify the expiration date on the package and do not use expired sets. Do not reuse single-use sets; each set is designed for one patient, one infusion episode, and must be discarded after use.
Priming and Air Elimination
Before connecting the IV set to a patient's vein, the entire length of tubing must be primed (filled with fluid) to remove air. Air remaining in the tubing can enter the bloodstream, creating an air embolism (in rare cases, a life-threatening condition). The standard priming procedure involves running fluid through the set into a waste container until no air bubbles remain in the tubing or Y-injection chamber. The clear tubing of the Exel set allows visual confirmation of complete priming.
Clamp Function and Flow Control Integrity
Both the roller clamp and occlusion clamp must function smoothly without resistance or grinding. A clamp that does not move or closes incompletely compromises the ability to regulate or stop flow. If a clamp is difficult to operate or does not fully compress the tubing, discard the set and use a new one. Attempting to force a stuck clamp risks tubing damage.
Y-Injection Site Asepsis
The Y-injection site is a portal for secondary medication administration. Each time the site is accessed with a needle, syringe, or secondary infusion line, there is a risk of introducing bacteria. Always disinfect the Y-site with an appropriate antiseptic (70% isopropyl alcohol or chlorhexidine) before each access. Allow the site to dry completely before injection or connection. Minimise the number of accesses and the duration the site remains exposed.
Catheter and Vein Monitoring
The IV set itself is only one component of infusion safety. The patient's peripheral or central catheter, the infusion site vein, and surrounding tissue must be monitored for signs of infiltration (fluid leaking into surrounding tissue), phlebitis (vein inflammation), or infection. A properly functioning IV set does not prevent these complications; appropriate site care, frequent monitoring, and timely catheter removal when no longer needed are the clinician's responsibilities.
Ordering, Professional Licensing, and Shipping Information
Professional Licensing Requirements
The Exel IV Administration Set is a medical device intended for use by licensed healthcare professionals. Mountainside Medical supplies this product to qualified buyers only, including:
- Licensed physicians, nurse practitioners, physician assistants, and nurses
- Hospitals, clinics, urgent care centers, and ambulatory surgery centers
- Emergency Medical Services (EMS) agencies and paramedic services
- Dental and oral surgery practices
- Veterinary clinics and animal hospitals
- Medical spas and aesthetics clinics with licensed practitioners
- Visiting nurse agencies and home health services
- Retail and institutional buyers with medical reseller licenses
- Law enforcement, fire departments, and emergency response organizations
At checkout, you will be asked to verify your professional license, facility credentials, or reseller authorization. Mountainside Medical reserves the right to request documentation of licensure or business registration before fulfilling orders. Providing false credentials or misrepresenting professional status may result in order cancellation and account suspension.
Individual consumers and non-licensed buyers cannot purchase prescription medications, controlled substances, or medical devices from Mountainside Medical. If you are a licensed healthcare professional ordering for personal or professional use, you must provide valid credentials.
New Customer Discount
First-time buyers receive 5% off their initial purchase. This discount is applied automatically at checkout for new customer accounts. No promotional code is required.
Pricing and Volume Discounts
Not specified in provided data. Contact Mountainside Medical directly for current pricing, volume discount tiers, and wholesale pricing for large orders or standing contracts:
- Phone: +1 (888) 687-4334
- Email: sales@mountainside-medical.com
Our sales team can provide custom quotes for:
- Bulk orders (50+ units or higher)
- Standing orders or monthly recurring supplies
- Annual contracts with volume-based pricing
- Facility-specific packaging or labeling requests
Shipping Information
Shipping Coverage: Free shipping on orders over $100 within the United States. Expedited shipping options are available; contact sales for rates and availability.
Specific delivery timelines for this product: Not specified in provided data. Upon order placement, you will receive an estimated delivery date based on current stock status and your location. Mountainside Medical prioritises fast order processing and reliable delivery as core service commitments for healthcare providers whose operations depend on consistent supply.
Stock Status: Not specified in provided data. To verify current availability of the Exel IV Administration Set before ordering, contact Mountainside Medical:
- Phone: +1 (888) 687-4334
- Email: sales@mountainside-medical.com
Order Modifications and Returns
Not specified in provided data. Contact Mountainside Medical directly regarding return policies, replacement procedures for damaged products, and order modification requests. Given the medical and time-sensitive nature of healthcare supply chains, communicate any issues immediately upon discovery.
Handling and Packaging
All Exel IV Administration Sets are shipped in sealed, sterile, single-use packaging. Upon receipt, inspect the outer shipping box and inner product packaging for damage. If packaging is opened, torn, or compromised during transit, do not use the product; contact Mountainside Medical for immediate replacement. Damaged or contaminated medical devices cannot be used on patients and must be documented and discarded per your facility's medical waste protocols.
Regulatory Compliance and Credentials
Mountainside Medical is committed to supplying medical devices and pharmaceuticals in compliance with federal regulations. Not specified in provided data: specific regulatory registrations (DEA number, pharmacy license, state medical device distributor licenses). For verification of Mountainside Medical's licensing and regulatory standing, contact the company directly or consult your state's pharmacy board or medical device distributor registry.
Contacting Mountainside Medical
- Phone: +1 (888) 687-4334
- Email: sales@mountainside-medical.com
- Website: https://www.mountainside-medical.com/
Our sales team is available to answer questions about product specifications, ordering processes, bulk pricing, shipping, licensing requirements, and any other aspect of your purchase.
Important Safety and Regulatory Information
Medical Device Disclaimer
The Exel IV Administration Set is a medical device intended for use by licensed healthcare professionals only. This product is not intended for self-administration, home use by untrained individuals, or any use outside the scope of licensed professional practice.
This product is NOT a substitute for professional medical advice, diagnosis, treatment, or clinical judgment. The information provided on this page is educational only and does not constitute medical advice. All decisions regarding patient care, infusion therapy, medication administration, and medical device selection must be made by qualified healthcare professionals based on individual patient assessment, clinical protocols, and facility policies.
Regulatory and Certification Status
Not specified in provided data: FDA device classification, specific ISO certifications, CE marking, or other regulatory clearances applicable to the Exel IV Administration Set. If you require verification of regulatory status, manufacturing standards, or certifications, contact Mountainside Medical or the product manufacturer directly.
Material Safety Claims
The Exel IV Administration Set is described as sterile, latex-free, and non-pyrogenic. These claims reflect product specifications as provided by the manufacturer. However, sterilization effectiveness, latex absence, and pyrogenic safety depend on proper storage, handling, and aseptic technique during use. Opening a sterile package, exposing the set to non-sterile conditions, or failing to follow standard aseptic protocol can compromise these safety properties.
Product Liability and Clinical Outcomes
Mountainside Medical and the Exel IV Administration Set manufacturer are not liable for patient outcomes, complications, or adverse events resulting from:
- Improper product selection or use outside recommended clinical applications
- Failure to follow aseptic technique or standard infection prevention protocols
- Use by unlicensed or untrained individuals
- Mishandling, improper storage, or use of expired products
- Use with incompatible equipment or solutions
- Clinician error or deviation from established clinical guidelines
- Patient-specific contraindications or complications unrelated to product defect
Reporting Adverse Events
If you experience any adverse event, product defect, or injury related to the Exel IV Administration Set, report it immediately to:
- Mountainside Medical: +1 (888) 687-4334 or sales@mountainside-medical.com
- FDA MedWatch Program: 1-888-SAFEMED (1-888-723-3633) or www.fda.gov/medwatch
Detailed reporting helps manufacturers and regulators identify product issues and protect patient safety across healthcare systems.
Professional Responsibility and Scope of Practice
Use of the Exel IV Administration Set falls within the scope of licensed professional practice. Users are responsible for:
- Understanding product specifications and appropriate clinical applications
- Following facility protocols and clinical practice guidelines
- Maintaining aseptic technique and infection prevention standards
- Monitoring infusion sites and patient responses during therapy
- Recognizing complications (infiltration, phlebitis, infection) and taking appropriate action
- Documenting infusion details per facility requirements
- Proper disposal of single-use medical devices per medical waste regulations
Changes to Product or Information
Mountainside Medical reserves the right to update product specifications, pricing, shipping policies, or other information on this page without notice. Check the website regularly for current information. If you have questions about product changes, contact Mountainside Medical directly.
Contact for Questions or Concerns
For any questions regarding safety, regulatory status, clinical applications, or concerns about this product, contact:
- Mountainside Medical
- Phone: +1 (888) 687-4334
- Email: sales@mountainside-medical.com
- Website: https://www.mountainside-medical.com/
Frequently Asked Questions: Exel IV Administration Set
What is the difference between a 60-drop IV set and a 10-drop IV set?
The number refers to the drop factor—how many drops equal one millilitre of fluid. A 60-drop (microdrip) set delivers 60 small drops per mL, while a 10-drop (macrodrip) set delivers 10 large drops per mL. The Exel 60-drop set provides more precise flow control because each drop represents a smaller volume. Microdrip sets are ideal for medications and low-volume infusions; macrodrip sets are faster and better for emergency fluid resuscitation. The choice depends on your clinical need: precision vs. volume delivery speed.
What does \"non-pyrogenic\" mean on an IV set?
Pyrogenic substances are endotoxins (bacterial fragments) that cause fever reactions in patients. A non-pyrogenic IV set has been manufactured and tested to eliminate or minimize these contaminants. The Exel set undergoes manufacturing processes (sterile filtration, steam sterilization, or gamma radiation) designed to remove pyrogenic substances before packaging. This safety assurance is important because fever during infusion therapy can complicate patient care and mask other medical conditions. Non-pyrogenic status, however, does not replace aseptic technique during insertion and infusion management.
Why is this IV set latex-free?
Natural rubber latex can trigger immediate or delayed allergic reactions in sensitive individuals. Symptoms range from mild (itching, localized swelling) to severe (anaphylaxis, breathing difficulty). By using latex-free materials throughout, the Exel set eliminates this risk for latex-allergic patients and clinicians. If a patient has a documented latex allergy, all connected equipment—IV bags, extension tubing, medication syringes, and secondary infusion sets—must also be latex-free to prevent a reaction.
What's the difference between Luer Lock and Luer Slip connectors?
Luer Lock features a threaded collar that twists onto the IV bag port, creating a secure, fixed connection resistant to accidental disconnection. Luer Slip is a straight cone that pushes onto the port with no threading. Luer Lock is preferred when connection security is critical (critical care, transport, high-movement scenarios); Luer Slip is faster for rapid setup and frequent disconnections (emergency departments, rapid-transfusion scenarios). When selecting your Exel set, match the connector type to your IV bags and workflow. Most standard IV bags accept both, but verify your specific container's port type if uncertain.
Can I use this IV set with any IV bag?
The Exel IV Administration Set uses standard luer connectors and is designed to be universal—compatible with any standard IV bag featuring a luer port. However, always verify: (1) your IV bag has a standard luer connector, and (2) the venting mode matches your bag type. Rigid plastic bags use non-vented operation (air cannot enter); glass bottles typically use vented operation. If your facility uses non-standard IV containers or bags without luer ports, contact the bag manufacturer or Mountainside Medical to confirm compatibility.
What does the Y-injection site do?
The Y-injection site is a secondary port that allows you to inject medications or infuse a secondary line without disconnecting the primary infusion. For example, if a patient is receiving a continuous medication infusion but needs a bolus dose of a different drug, you can inject the bolus through the Y-site while the primary infusion continues. This reduces manipulation and disconnection of the main line, lowering infection risk and maintaining continuous therapy. Always disinfect the Y-site with 70% isopropyl alcohol before each access to prevent introducing bacteria.
What does the occlusion clamp do, and why have two clamps?
Both the roller clamp and occlusion clamp shut off fluid flow, but they work differently. The roller clamp compresses the flexible tubing to restrict flow; the occlusion clamp fully closes the tubing. The dual-clamp design provides redundancy and flexibility: use the roller clamp for fine flow adjustment, and the occlusion clamp for complete shutdown without compressing the tubing (useful during dressing changes or patient repositioning). Some clinicians use the occlusion clamp as a safety backup if the roller clamp malfunctions.
How do I prime an IV set and remove air?
Priming fills the entire tubing with fluid and removes air bubbles before connecting to a patient. Procedure: (1) Attach the primary connector to your IV bag. (2) Open the roller clamp and allow fluid to run freely through the tubing into a waste container. (3) Watch the clear tubing closely—the fluid should move continuously, pushing air out. (4) When fluid reaches the Y-injection chamber and all air is gone from the tubing, the set is primed. (5) Close the roller clamp and connect to the patient's IV catheter. The clear tubing of the Exel set allows visual confirmation of complete priming, reducing the risk of air entering the bloodstream.
What does the integrated valve (vented/non-vented option) do?
The Exel IV set includes a valve that switches between vented and non-vented operation. In vented mode, air enters the IV bag as fluid drains, allowing continuous flow (used with glass bottles). In non-vented mode, air is blocked, preventing backflow and keeping the infusion closed (used with rigid plastic bags). Before connecting your set, check your IV bag type. Mismatching venting to bag type causes flow to slow, stop, or create backflow. If unsure, your facility's IV protocol or the IV bag packaging will specify which mode is required.
Can the Exel IV set be reused?
No. The Exel IV Administration Set is a single-use, disposable medical device. Each set is designed for one patient and one infusion episode only. After use, discard the set per your facility's medical waste disposal protocols. Reusing or attempting to resterilize a single-use IV set is unsafe—the tubing and components cannot be reliably re-sterilized, and repeated use increases contamination risk.
What if the IV set is damaged or the packaging is opened?
Do not use a damaged or compromised IV set. If the packaging is torn, opened, or shows signs of contamination, or if you see cracks, holes, or cloudiness in the tubing, discard the set immediately and use a new, unopened product. Damaged equipment cannot maintain sterility and poses a patient safety risk. If you receive a damaged product, contact Mountainside Medical at +1 (888) 687-4334 or sales@mountainside-medical.com for immediate replacement.
How should I store Exel IV sets?
Store in a dry location at room temperature (68–77°F / 20–25°C), protected from direct sunlight and extreme heat. Heat can degrade the tubing material. Check the expiration date on the package; most IV sets have a 3–5 year shelf life from the manufacturing date. Do not use expired products. If stored properly in sealed, unopened packaging, the set will remain sterile until the expiration date.
Who should not use this IV set?
The Exel 60-drop IV set is not appropriate for: (1) Emergency rapid infusion or massive fluid resuscitation (use macrodrip 10-drop sets instead), (2) Unlicensed or untrained individuals (professional use only), (3) Patients with non-standard or incompatible IV equipment, (4) Non-Luer-port IV containers. Additionally, while the set is latex-free, patients with other component allergies (PVC, silicone, plasticizers) may require verification of all tubing materials before use. If you are uncertain whether the Exel set is appropriate for a specific patient or clinical situation, consult your facility's clinical protocols or contact a supervisor.
How do I order the Exel IV Administration Set?
Visit Mountainside Medical online at https://www.mountainside-medical.com/, search for \"Exel IV Administration Set,\" and add to your cart. You will be required to verify your professional credentials or facility licensing at checkout. First-time customers receive 5% off. Free shipping applies to orders over $100 within the United States. For bulk orders, volume discounts, or questions about pricing, contact Mountainside Medical: Phone: +1 (888) 687-4334 or Email: sales@mountainside-medical.com.
What are the shipping and delivery options?
Free shipping on orders over $100 within the US. Specific delivery timelines are not specified in the provided data; upon order placement, you will receive an estimated delivery date. Expedited shipping options may be available—contact Mountainside Medical at +1 (888) 687-4334 for rates and availability. For questions about delivery status or estimated arrival, contact the sales team via phone or email.
What if I have questions or experience problems with the product?
Contact Mountainside Medical immediately: Phone: +1 (888) 687-4334, Email: sales@mountainside-medical.com, or Website: https://www.mountainside-medical.com/. For product defects, adverse reactions, or safety concerns, also report to the FDA MedWatch Program: 1-888-SAFEMED (1-888-723-3633) or www.fda.gov/medwatch. Prompt reporting helps manufacturers and regulators protect patient safety.