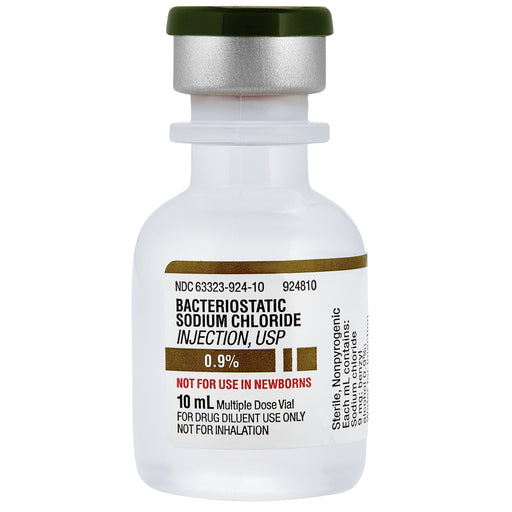
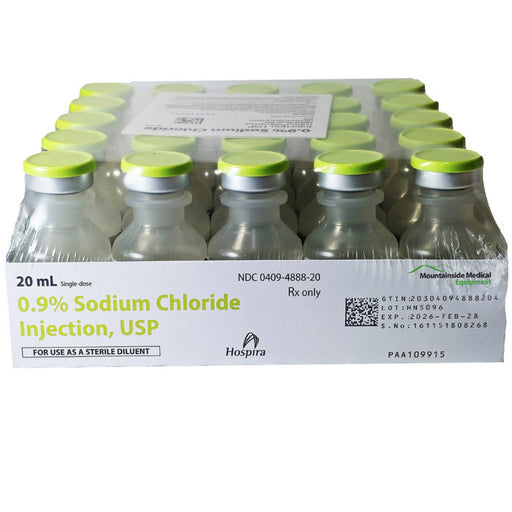
Bacteriostatic Sodium Chloride 0.9% Injection Multiple Dose Vials are used as a diluent for medications, providing a sterile solution for injection. They contain benzyl alcohol as a preservative to prevent bacterial growth, allowing multi-dose usage. This solution is employed for diluting or dissolving medications for intravenous, intramuscular, or subcutaneous administration. It helps maintain the isotonicity of drug solutions, ensuring safe medication delivery. Commonly used in clinical settings, it aids in administering consistent and effective treatment across various medical applications.
Details and Features:
Volume: 20 mL per vial
Packaging: 25 vials per pack
Concentration: 0.9% Sodium Chloride
Preservative: Contains benzyl alcohol (0.9%) as a bacteriostatic agent
Intended Use: Diluting or dissolving medications for injection
Multiple-dose vial: Allows multiple withdrawals for various dilutions or preparations
Mechanism of Action:
The primary role of Bacteriostatic Sodium Chloride 0.9% Injection is to act as a vehicle for the administration of medications. It provides a sterile solution in which drugs can be diluted without compromising their efficacy. The sodium chloride provides isotonicity, making it compatible with the body's extracellular fluids. The added benzyl alcohol prevents bacterial growth, thus permitting multiple uses from the same vial under appropriate conditions.
Warnings:
-
Not For Direct Intravenous Use: This product must not be administered intravenously without proper dilution with other compatible medications, as it is intended solely as a diluent.
-
Benzyl Alcohol Warning: The presence of benzyl alcohol can cause serious side effects in certain populations, such as neonates, and should be used cautiously in pregnant women or individuals with known allergies to benzyl alcohol.
-
Consultation Required: Only use as directed by a healthcare provider and ensure compatibility with the medication to be diluted.
-
Reuse Limitation: Intended for multiple use by a single individual, ensuring sterility is maintained during each access to the product. Always wipe the rubber stopper with an antiseptic before puncture.
-
Storage: Maintain in a suitable environment, protecting it from light and freezing.
Side Effects:
- The solution itself is typically well-tolerated when used as directed for preparation, but side effects can arise from:
-
Allergic Reactions: Potential hypersensitivity to ingredients, including benzyl alcohol.
-
Local Reactions: Site irritation could occur if the product is improperly administered.
-
Systemic Issues: If used in large quantities due to benzyl alcohol, it could cause metabolic complications in vulnerable groups, such as infants ("gasping syndrome").
Always ensure that the product is used in line with medical guidance and drilled protocol to prevent adverse effects connected to its administration.