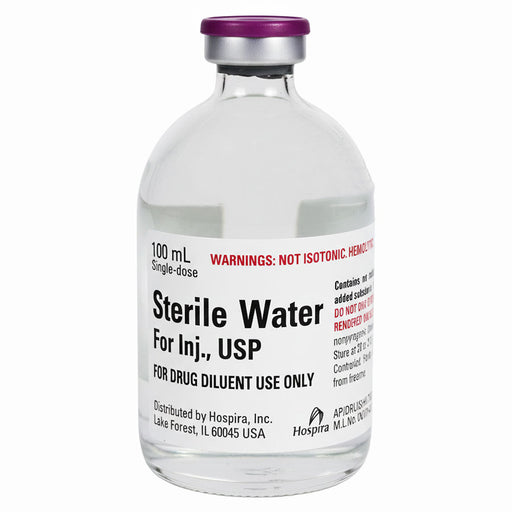
Bacteriostatic Water for Injection is a pharmaceutical-grade sterile diluent formulated with 0.9% benzyl alcohol as a preservative, enabling safe multiple withdrawals from a single vial without contamination when aseptic technique is maintained. Manufactured by Hospira Inc. (a Pfizer subsidiary) and meeting USP monograph standards, this product is the industry standard for medication reconstitution, peptide preparation, and injectable therapy across hospitals, clinics, compounding pharmacies, and licensed aesthetic practices.
This product is available exclusively for licensed healthcare professionals and institutions. Mountainside Medical offers competitive wholesale pricing, fast US-wide shipping (free over $100), and direct access to the trusted Hospira Pfizer formulation that practitioners rely on daily.
Wholesale pricing with fast US shipping over $100 | New customers receive 5% off first purchase | Rx-only product for licensed healthcare professionals
WHAT IS BACTERIOSTATIC WATER
Bacteriostatic Water for Injection is a sterile, non-pyrogenic solution of water containing 0.9% benzyl alcohol as a preservative. The benzyl alcohol acts as a bacteriostatic agent — inhibiting bacterial growth without being bactericidal. This distinction is critical: the preserved formula allows repeated doses to be drawn from the same multi-dose vial without risk of contamination, provided proper aseptic technique is maintained.
Unlike regular sterile water, which requires single-dose use and immediate discarding after opening, bacteriostatic water can safely remain in a vial for extended periods. The product appears as a clear, colourless solution with no visible particles. Each vial is sealed in accordance with pharmaceutical manufacturing standards and tested for sterility before distribution.
| Attribute |
Specification |
| Product Name |
Bacteriostatic Water for Injection, USP |
| Manufacturer |
Hospira Inc. (Pfizer subsidiary) |
| NDC Code |
0409-3977-03 |
| Active Ingredient |
Sterile Water USP with 0.9% Benzyl Alcohol (preservative) |
| Volume per Vial |
30 mL |
| Pack Size |
25 multi-dose vials per pack |
| Regulatory Status |
USP Grade, FDA Approved, cGMP Manufactured |
| Administration Routes |
Intravenous (IV), Intramuscular (IM), Subcutaneous (SC), Intradermal (ID) |
| Multi-Dose Vial |
Yes — safe for repeated use with proper aseptic technique |
| Availability |
Rx-only; licensed healthcare professionals only |
Bacteriostatic Water for Injection USP — Overview
Bacteriostatic Water for Injection is a sterile, non-pyrogenic solution manufactured by Hospira Inc., a Pfizer subsidiary, and formulated with 0.9% benzyl alcohol as a preservative. This pharmaceutical-grade diluent is the industry standard for medication reconstitution, peptide preparation, and injectable therapies across hospitals, clinics, compounding pharmacies, and licensed aesthetic practices.
Unlike regular single-dose sterile water, which requires immediate use and disposal after opening, bacteriostatic water contains a preservative that inhibits bacterial growth. This critical distinction allows repeated doses to be drawn safely from the same multi-dose vial without risk of contamination, provided proper aseptic technique is maintained.
What Makes Bacteriostatic Water Different?
The benzyl alcohol in bacteriostatic water acts as a bacteriostatic agent—meaning it slows or prevents bacterial proliferation without destroying microorganisms (bactericidal action). This subtle but important mechanism enables the solution to remain stable and safe for extended periods within a sealed vial when aseptic handling practices are followed.
In clinical and aesthetic settings, this preservative-based formula translates to operational efficiency: fewer vials opened, less product waste, and reduced cost per dose for high-volume users. A single 30mL multi-dose vial can support multiple medication preparations or injections over time, making it particularly valuable for institutions and busy practices.
Product Appearance and Quality Standards
The solution appears as a clear, colourless liquid with no visible particles. Every vial is sealed according to pharmaceutical manufacturing standards and tested for sterility before distribution. Manufactured under cGMP (current Good Manufacturing Practice) conditions and meeting United States Pharmacopoeia (USP) monograph standards, this product carries FDA approval and is classified as prescription-only for licensed healthcare professionals.
Why Hospira Pfizer?
Hospira, now part of Pfizer, is recognised globally as a trusted manufacturer of pharmaceutical injectables and critical care solutions. This product represents decades of manufacturing expertise in sterile formulations, quality control, and regulatory compliance. Healthcare institutions and licensed practitioners rely on Hospira bacteriostatic water as the trusted foundation for medication delivery systems.
Key Facts at a Glance
- Manufacturer: Hospira Inc. (Pfizer subsidiary)
- Active Ingredient: Sterile Water USP with 0.9% Benzyl Alcohol
- Volume per Vial: 30 mL
- Pack Size: 25 multi-dose vials
- NDC Code: 0409-3977-03
- Regulatory Status: USP Grade, FDA Approved, cGMP Manufactured
- Availability: Rx-only; licensed healthcare professionals only
- Wholesale Pricing: Competitive rates with fast US shipping (free over $100)
- New Customer Benefit: 5% discount on first purchase
Common Applications in Professional Settings
Bacteriostatic water serves as essential infrastructure across multiple clinical and therapeutic environments:
- Medication Reconstitution: Dissolving lyophilised (freeze-dried) powders to prepare injectable medications
- Peptide Preparation: Diluting research or therapeutic peptide compounds for injection
- Injectable Therapy Support: Base solution for custom medication formulations in hospitals and clinics
- Compounding Pharmacy Use: Standard diluent for institutional pharmacy reconstitution protocols
- Aesthetic and Medical Practice: Diluent for injectable treatments in licensed medical and aesthetic settings
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Complete Product Specifications
| Attribute |
Specification |
| Product Name |
Bacteriostatic Water for Injection, USP |
| Manufacturer |
Hospira Inc. (Pfizer subsidiary) |
| NDC Code |
0409-3977-03 |
| Active Ingredient |
Sterile Water USP with 0.9% Benzyl Alcohol (preservative) |
| Volume per Vial |
30 mL |
| Pack Size |
25 multi-dose vials per pack |
| Vial Type |
Multi-dose vial (safe for repeated use with proper aseptic technique) |
| Solution Appearance |
Clear, colorless, no visible particles |
| Sterility Status |
Sterile, non-pyrogenic; tested for sterility before distribution |
| Regulatory Status |
USP Grade, FDA Approved, cGMP Manufactured |
| Administration Routes |
Intravenous (IV), Intramuscular (IM), Subcutaneous (SC), Intradermal (ID) |
| Preservative Mechanism |
Benzyl alcohol (bacteriostatic agent; inhibits bacterial growth) |
| Prescription Status |
Rx-only; available exclusively to licensed healthcare professionals and institutions |
Multi-Dose Vial Safety & Extended Shelf-Life
The 0.9% benzyl alcohol preservative is the key functional difference between bacteriostatic water and single-dose sterile water. This concentration is specifically selected to meet USP monograph requirements for safety and efficacy in multi-dose vial applications.
When stored properly and accessed using aseptic technique, a single 30mL vial can support multiple withdrawals over an extended period. This extends the practical utility of each vial compared to single-dose formulations that must be discarded immediately after opening.
Manufacturing & Quality Assurance
This product is manufactured under cGMP (current Good Manufacturing Practice) standards, which means the facility, equipment, personnel, and processes meet strict FDA regulatory requirements. Every batch undergoes sterility testing and quality control verification before release. Hospira's manufacturing heritage ensures consistent, reliable formulation across all units.
Clinical and Professional Applications
Bacteriostatic Water in Medication Reconstitution
Bacteriostatic water is the standard diluent for reconstituting lyophilized (freeze-dried) medications in clinical and institutional settings. Many injectable drugs are manufactured in powder form to extend shelf-life. Before administration, these powders must be dissolved in a sterile diluent—and bacteriostatic water is the preferred choice for medications requiring multi-dose vial stability.
When a medication powder is reconstituted with bacteriostatic water, the benzyl alcohol preservative continues to protect the solution against microbial contamination. This means a single reconstituted vial can be accessed multiple times over hours or even days, reducing waste and improving operational efficiency in busy clinical environments.
Peptide Preparation and Research Use
Licensed healthcare practitioners, compounding pharmacies, and research institutions use bacteriostatic water to prepare peptide injections. Peptides—short chains of amino acids—are increasingly used in therapeutic and aesthetic applications. Reconstituting peptide powders with bacteriostatic water preserves solution integrity and allows for staged use of a single vial.
This application requires strict adherence to aseptic technique and is available exclusively to licensed professionals and institutional buyers.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Injectable Therapy in Institutional Settings
Hospitals, surgical centers, and clinics rely on bacteriostatic water as foundational infrastructure for injectable treatments. Whether for IV medication administration, intramuscular injections, subcutaneous therapy, or intradermal procedures, this diluent supports safe, sterile preparation and delivery.
The multi-dose capability is particularly valuable in high-volume settings where multiple patients may require the same medication. A single vial can service several doses or procedures, reducing procurement burden and storage space requirements.
Aesthetic and Medical Practice Applications
Licensed aesthetic professionals, medical spas, dental practices, and medical offices use bacteriostatic water for injectable cosmetic and therapeutic procedures. In these settings, bacteriostatic water serves as the diluent for injectable treatments administered under professional supervision.
The preservative-based formulation allows practitioners to prepare multiple doses from a single vial during a treatment session or across multiple appointments, improving workflow efficiency and cost management.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
Compounding Pharmacy Standard
Compounding pharmacies use bacteriostatic water as the standard diluent for preparing custom injectable formulations. Institutional and retail pharmacies rely on this USP-grade solution to meet quality and safety standards when reconstituting medications or preparing sterile injections for patients.
EMS and Emergency Response
Emergency Medical Services (EMS) and emergency responders maintain bacteriostatic water in their drug preparation and medication administration protocols. Its multi-dose vial format supports rapid medication preparation in field and clinical emergency situations.
Visiting Nurse and Home Care Services
Visiting nurse agencies and home healthcare providers use bacteriostatic water for patient medication administration. The multi-dose vial format reduces waste and supports cost-effective care delivery across multiple patient visits.
Veterinary and Dental Practice
Licensed veterinary and dental practices use bacteriostatic water for injectable medication preparation and animal patient care. The same multi-dose vial benefits apply in these specialized settings.
Safety Information and Proper Use
Aseptic Technique: The Critical Foundation for Multi-Dose Safety
The safety of bacteriostatic water in multi-dose vials depends entirely on aseptic technique—the use of sterile equipment and procedures to prevent contamination. Despite the benzyl alcohol preservative, improper handling can introduce bacteria or other microorganisms that may overwhelm the preservative's protective capacity.
Proper aseptic technique includes:
- Cleaning the vial septum with appropriate antiseptic (typically 70% isopropyl alcohol) before each access
- Using a sterile needle and syringe for each withdrawal
- Maintaining sterile conditions throughout the preparation process
- Avoiding touch contamination of the vial septum or needle
- Following institutional or professional protocols for multi-dose vial management
Consequence of Improper Technique: Failure to maintain aseptic technique can result in microbial contamination of the vial contents, potentially leading to serious infection if contaminated solution is administered. Healthcare professionals must understand and consistently apply aseptic principles when using multi-dose vials.
Benzyl Alcohol Considerations
While benzyl alcohol is an FDA-approved preservative for multi-dose vial formulations, it is important to note that:
- Benzyl alcohol has been associated with adverse effects in neonates (very young infants) at high concentrations when used as a preservative in injectable products
- Use in pediatric or neonatal populations should follow institutional guidelines and medical supervision
- Practitioners should be aware of any patient-specific contraindications or sensitivities
- Product use should always align with the prescribing information and clinical judgment of licensed healthcare providers
Storage and Handling Requirements
Temperature: Store at controlled room temperature (typically 68–77°F / 20–25°C). Do not freeze or expose to extreme heat.
Light Protection: Protect from direct sunlight and strong light exposure.
Shelf-Life: This product has a defined expiration date printed on the vial. Do not use after expiration.
Opened Vial Duration: Once opened and accessed via needle puncture, the vial remains viable only if aseptic technique is maintained and storage conditions are met. Follow institutional protocols or product labeling for guidance on maximum duration of use after opening (typically ranging from hours to days depending on clinical setting and protocol).
Inspection Before Use
Before using any vial, visually inspect for:
- Cloudiness or discoloration (solution should be clear and colorless)
- Visible particles or precipitate
- Cracks, leaks, or damage to the vial or septum
- Any sign of contamination or compromise
Do not use vials that show any sign of contamination or damage. Discard appropriately according to institutional hazardous waste protocols.
Prescription-Only Status and Licensing Requirements
This product is available exclusively to licensed healthcare professionals, licensed institutions (hospitals, clinics, pharmacies, EMS agencies), and authorized distributors. It is not available for retail purchase at pharmacies, online retailers targeting consumers, or over-the-counter outlets.
Why Rx-Only? Bacteriostatic water is a regulated pharmaceutical product that requires professional training in aseptic technique, proper storage, and safe handling to prevent harm. Its multi-dose vial format and benzyl alcohol preservative require understanding of microbiology and infection control principles that are beyond the scope of consumer use.
Adverse Effects and Reporting
Adverse events or suspected contamination issues should be reported immediately to the manufacturer (Hospira/Pfizer) and relevant regulatory authorities (FDA MedWatch, poison control, or institutional pharmacovigilance systems). Licensed healthcare professionals are trained in adverse event recognition and reporting protocols.
Contraindications and Precautions
Use of bacteriostatic water should be guided by:
- The prescribing information of the medication being reconstituted
- Institutional policies and clinical protocols
- Professional guidelines (pharmacy boards, nursing associations, medical societies)
- Individual patient factors and medical history (as determined by the treating healthcare provider)
Practitioners should verify that bacteriostatic water is the appropriate diluent for the specific medication being reconstituted, as some medications may require single-dose sterile water or other specific diluents.
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
How to Order Bacteriostatic Water Wholesale
Licensing Requirements
Bacteriostatic Water for Injection is a prescription-only pharmaceutical product. To purchase from Mountainside Medical, you must be:
- A licensed healthcare professional: Licensed physician, nurse practitioner, pharmacist, physician assistant, dentist, veterinarian, or other healthcare provider with active licensure in your state
- An authorized institutional buyer: Hospital, surgical center, clinic, compounding pharmacy, EMS agency, fire department, or other licensed institutional healthcare facility
- An authorized distributor or medical wholesale account: Current business license and DEA registration (if applicable to your state regulations)
At checkout or during account setup, you will be asked to verify your professional license or institutional authorization. Mountainside Medical maintains records of all purchases for regulatory compliance and can request verification documentation at any time.
Why We Verify Licensing
Verification ensures compliance with federal and state pharmacy regulations and protects the integrity of the pharmaceutical supply chain. It also ensures the product reaches professionals who have the training and protocols in place to handle it safely.
Individual Professional Orders
For licensed practitioners (physicians, dentists, veterinarians, nurses, pharmacists, etc.):
- Visit https://www.mountainside-medical.com/products/bacteriostatic-water-injection-pfizer-injectables
- Add the 30mL 25-pack (NDC 0409-3977-03) to your cart
- Proceed to checkout
- Provide your professional license number or institutional affiliation for verification
- Select shipping and payment method
- Place your order
New customers receive 5% off your first purchase.
Institutional and Bulk Orders
For hospitals, clinics, compounding pharmacies, EMS agencies, and other institutions:
For bulk orders, volume pricing, or custom account setup, contact our sales team directly:
Phone: +1 (888) 687-4334
Email: sales@mountainside-medical.com
Our wholesale team will:
- Verify your institutional license and DEA registration (if applicable)
- Discuss your volume needs and usage patterns
- Provide competitive wholesale pricing and volume discounts
- Set up a standing order or blanket purchase agreement if needed
- Ensure fast, reliable delivery on your schedule
Shipping & Delivery
Coverage Area: Continental United States (all 50 states)
Shipping Cost: FREE on orders over $100 | Standard shipping available for smaller orders
Delivery Timeline: Fast US shipping with expedited options available. Orders typically ship within 1–2 business days of placement.
Packaging: All pharmaceutical products are shipped in secure, climate-controlled packaging to ensure product integrity and regulatory compliance during transit.
Tracking: All orders include tracking information so you can monitor delivery status in real-time.
Why Choose Mountainside Medical?
- Trusted Manufacturer: Direct access to Hospira Pfizer formulation
- Competitive Wholesale Pricing: Better per-unit cost than retail channels
- Fast, Reliable Delivery: Free shipping on orders over $100; expedited options available
- New Customer Incentive: 5% off your first purchase
- Expert Customer Service: Licensed healthcare professionals available to answer questions
- Regulatory Compliance: Full licensing verification and documentation for institutional compliance
- Bulk & Standing Order Support: Streamlined procurement for high-volume users
Questions Before Ordering?
Contact our sales team:
Phone: +1 (888) 687-4334
Email: sales@mountainside-medical.com
We can clarify licensing requirements, discuss bulk options, answer product questions, or help you with any aspect of the ordering process.
Why Bacteriostatic Water Is Not Available at Retail Pharmacies
You may have searched for bacteriostatic water at CVS, Walgreens, or other retail pharmacies only to find it unavailable. This is not an oversight—it is by regulatory design.
Bacteriostatic water is a prescription pharmaceutical product intended exclusively for professional and institutional use. Retail pharmacies do not stock it because:
- Rx-Only Status: The FDA classifies this product as prescription-only, meaning it requires professional licensure and training to use safely
- Professional Use Only: The multi-dose vial format and benzyl alcohol preservative require understanding of aseptic technique and infection control—knowledge beyond consumer scope
- Wholesale Distribution Model: Manufacturers like Hospira distribute through licensed wholesale channels (medical wholesalers, authorized distributors) rather than retail pharmacy networks
- Regulatory Compliance: Supply chain oversight ensures products reach qualified professionals with proper training and protocols
The Solution: If you are a licensed healthcare professional, order directly from Mountainside Medical at competitive wholesale prices. We specialize in serving practitioners and institutions with professional-grade medical supplies and pharmaceuticals.
Product Disclaimer & Important Information
Prescription-Only Product Notice
Bacteriostatic Water for Injection is a prescription-only pharmaceutical product. It is available exclusively to licensed healthcare professionals, authorized institutions, and registered distributors. By purchasing this product, you confirm that you hold valid professional licensure or institutional authorization to procure pharmaceutical products.
Professional Use Only
This product is intended for use by qualified healthcare professionals in clinical, institutional, and professional settings only. It is not intended for consumer purchase, self-administration, or use without proper professional supervision and training in aseptic technique and pharmaceutical handling.
Medical Guidance Disclaimer
The information provided on this product page is for educational purposes for licensed healthcare professionals and authorized institutional buyers. It is not a substitute for professional medical advice, diagnosis, or treatment. Licensed practitioners must:
- Consult the product's full prescribing information and the prescribing information of any medication being reconstituted
- Follow all applicable federal, state, and local pharmaceutical regulations
- Adhere to institutional protocols and professional guidelines relevant to their practice
- Exercise independent professional judgment in determining the appropriateness of this product for their specific use case
Safety and Aseptic Technique
Safe use of bacteriostatic water in multi-dose vials depends entirely on proper aseptic technique and handling. Failure to maintain aseptic conditions can result in microbial contamination and serious infection. Users must be trained in and consistently apply:
- Proper vial septum disinfection procedures
- Sterile needle and syringe use
- Aseptic vial access and withdrawal techniques
- Appropriate storage and handling
Mountainside Medical is not responsible for improper use, storage, handling, or violations of aseptic technique by end users.
Product Verification
This product (Bacteriostatic Water for Injection USP, Hospira, NDC 0409-3977-03) is manufactured by Hospira Inc., a Pfizer subsidiary, and is subject to FDA oversight and USP standards. Mountainside Medical is an authorized distributor and does not manufacture or reformulate this product. The product you receive will be the authentic Hospira formulation as described in this listing.
No Medical Claims
This product page does not claim that bacteriostatic water treats, cures, prevents, or mitigates any disease or medical condition. It is a pharmaceutical diluent intended for medication reconstitution and injectable therapy preparation under professional supervision. Any clinical benefit or outcome depends on the medication being reconstituted, the patient's medical condition, and the professional judgment of the treating healthcare provider.
Liability Limitation
Mountainside Medical provides this product in accordance with all applicable federal and state pharmaceutical laws. Users are responsible for:
- Verifying their own licensure and authorization to purchase this product
- Understanding and complying with all federal, state, and local regulations governing pharmaceutical use and handling
- Following proper aseptic technique and institutional protocols
- Seeking appropriate medical or pharmaceutical guidance for clinical questions
Mountainside Medical disclaims liability for injury, infection, or adverse outcomes resulting from improper use, storage, handling, or violation of aseptic technique by users or institutions.
Storage and Expiration
Store this product according to the manufacturer's instructions (typically room temperature, protected from freezing, heat, and light). Check the expiration date on the vial. Do not use after expiration. Opened vials must be used and stored according to institutional protocols and product guidance.
Reporting Adverse Events or Safety Concerns
If you experience or suspect an adverse event, contamination, or quality issue with this product, report it immediately to:
- Hospira Pfizer: Manufacturer contact information available on the product vial
- FDA MedWatch: 1-888-SAFEMED (1-888-723-3633) or online at fda.gov/medwatch
- Poison Control: 1-800-222-1222 (for toxicology guidance)
- Your institutional pharmacovigilance system: If you work for a hospital, clinic, or healthcare institution
Regulatory Compliance
By purchasing and using this product, you agree to comply with all applicable FDA regulations, state pharmacy laws, and professional licensing requirements. Mountainside Medical maintains records of all sales for regulatory compliance and may be required to provide documentation to regulatory authorities.
Contact for Regulatory or Compliance Questions
If you have questions about regulatory status, compliance, or the appropriateness of this product for your setting, contact:
Phone: +1 (888) 687-4334
Email: sales@mountainside-medical.com
This disclaimer was last updated in accordance with FDA and USP standards. It may be updated at any time to reflect regulatory changes or safety information.
Frequently Asked Questions
Product & Usage Questions
Q: What is bacteriostatic water used for?
A: Bacteriostatic water is used as a sterile diluent for medication reconstitution, peptide preparation, and injectable therapies in clinical and aesthetic settings. It dissolves powdered medications and allows safe repeated use from a single multi-dose vial when proper aseptic technique is maintained.
Q: How is bacteriostatic water different from regular sterile water?
A: Regular sterile water is single-dose only—it must be discarded immediately after opening. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth and allows safe multiple withdrawals from the same vial over time.
Q: What does the benzyl alcohol preservative do?
A: Benzyl alcohol is a bacteriostatic agent, meaning it slows or prevents bacterial growth without destroying microorganisms. This preservative keeps the solution sterile and safe for extended use from a multi-dose vial, provided aseptic technique is followed.
Q: Can I use bacteriostatic water for any medication?
A: No. You must verify that bacteriostatic water is the appropriate diluent for the specific medication you are reconstituting. Some medications may require single-dose sterile water or other specific diluents. Always check the medication's prescribing information or consult with your pharmacy.
Q: What routes of administration does bacteriostatic water support?
A: This product is formulated for intravenous (IV), intramuscular (IM), subcutaneous (SC), and intradermal (ID) administration routes.
Safety & Handling Questions
Q: How long can I use a vial of bacteriostatic water after opening it?
A: Duration depends on proper storage conditions and aseptic technique. Follow your institutional protocols or product labeling guidance. Typical durations range from hours to days. Always inspect for contamination before use.
Q: What is aseptic technique, and why is it so important?
A: Aseptic technique is the use of sterile equipment and procedures to prevent contamination. It includes cleaning the vial septum with alcohol before each access, using sterile needles, and maintaining clean conditions. Improper aseptic technique can introduce bacteria that may overwhelm the preservative, leading to serious infection if contaminated solution is administered.
Q: How should I store bacteriostatic water?
A: Store at controlled room temperature (68–77°F / 20–25°C). Protect from freezing, extreme heat, and direct sunlight. Check the expiration date on the vial and do not use after expiration.
Q: What should I do if I find visible particles or cloudiness in a vial?
A: Do not use the vial. Discard it according to your institutional hazardous waste protocol and report the issue to Mountainside Medical or the manufacturer.
Q: Are there any patient populations for whom bacteriostatic water is not appropriate?
A: Benzyl alcohol has been associated with adverse effects in neonates at high concentrations. Use in pediatric or neonatal populations should follow institutional guidelines and medical supervision. Consult with the treating healthcare provider regarding any patient-specific contraindications.
Regulatory & Licensing Questions
Q: Do I need a prescription for bacteriostatic water?
A: Yes, bacteriostatic water is a prescription-only pharmaceutical product. It is available exclusively to licensed healthcare professionals and authorized institutions.
Q: Why is bacteriostatic water Rx-only?
A: The multi-dose vial format and benzyl alcohol preservative require professional training in aseptic technique, proper storage, and infection control. The FDA restricts this product to professionals with the knowledge and infrastructure to use it safely.
Q: Can I buy bacteriostatic water at CVS, Walgreens, or other retail pharmacies?
A: No. Bacteriostatic water is not available at retail pharmacy chains. It is distributed exclusively through pharmaceutical wholesalers and authorized professional distributors like Mountainside Medical.
Q: Can I buy bacteriostatic water over the counter?
A: No. Bacteriostatic water is a prescription pharmaceutical product and is not available over-the-counter.
Q: What proof of licensure do I need to provide to order from Mountainside Medical?
A: During account setup or checkout, you will provide your professional license number or institutional credentials. Mountainside Medical verifies licensing information to ensure regulatory compliance.
Q: Who can buy bacteriostatic water?
A: Licensed healthcare professionals (physicians, nurses, pharmacists, dentists, veterinarians, PAs, NPs, etc.), authorized healthcare institutions (hospitals, clinics, compounding pharmacies, EMS agencies), and licensed distributors can purchase from Mountainside Medical.
Ordering & Shipping Questions
Q: What is the product code (NDC) for this bacteriostatic water?
A: NDC 0409-3977-03 — Hospira Bacteriostatic Water for Injection USP, 30mL, 25-pack.
Q: Do you offer bulk or case discounts?
A: Yes. Contact our sales team at +1 (888) 687-4334 or sales@mountainside-medical.com for wholesale pricing on bulk orders, volume discounts, and standing purchase agreements.
Q: How fast can you ship?
A: Orders typically ship within 1–2 business days. We offer standard and expedited shipping options. All orders over $100 include free shipping to the continental United States.
Q: Do you ship to all 50 states?
A: Yes, we ship free over $100 to all 50 US states. Some products may have state-specific restrictions; verify during checkout.
Q: What is the new customer discount?
A: New customers receive 5% off their first purchase.
Q: Can I set up a standing order or blanket purchase agreement?
A: Yes, especially for institutional buyers. Contact our sales team to discuss your volume needs and set up customized ordering arrangements.
Q: How do I track my order?
A: All orders include tracking information. You will receive a tracking number via email after your order ships.
Manufacturer & Product Questions
Q: Is this the same bacteriostatic water used in hospitals?
A: Yes. This Hospira Pfizer formulation (NDC 0409-3977-03) is the same pharmaceutical-grade product used in hospitals, clinics, compounding pharmacies, and institutional settings nationwide.
Q: Why Hospira Pfizer?
A: Hospira, now part of Pfizer, is a globally recognized manufacturer of sterile pharmaceutical injectables. This product meets USP standards, is FDA-approved, and is manufactured under cGMP (current Good Manufacturing Practice) conditions.
Q: Is this product FDA-approved?
A: Yes. This product is FDA-approved and manufactured to meet United States Pharmacopeia (USP) monograph standards and current Good Manufacturing Practice (cGMP) requirements.
Q: What countries is bacteriostatic water available in?
A: Mountainside Medical serves the United States. International availability depends on local pharmaceutical regulations and licensing requirements in other countries.
Questions Not Answered Here?
Contact our team:
Phone: +1 (888) 687-4334
Email: sales@mountainside-medical.com