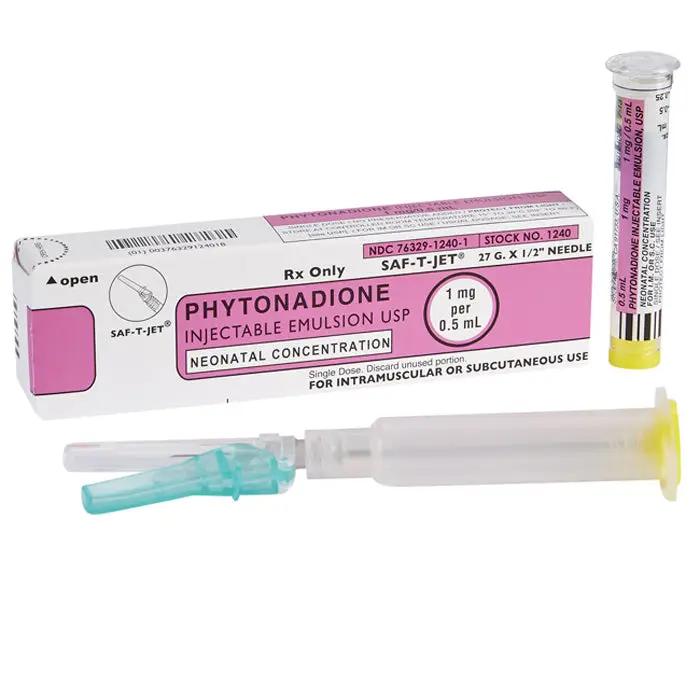
Phytonadione Injectable Emulsion Prefilled Syringes Neonatal Concentration 1 mg Per 0.5 mL by International Medication Systems refers to a medication that contains the active ingredient phytonadione, also known as vitamin K1. This medication is specifically designed for use in newborns and is provided in pre-filled syringes with a concentration of 1 mg per 0.5 mL. These syringes are produced by International Medication Systems, a pharmaceutical company. Phytonadione is used to prevent and treat bleeding disorders in newborns, particularly those caused by a vitamin K deficiency.
Phytonadione is a synthetic form of vitamin K, which is important for blood clotting. In neonatal patients, vitamin K deficiency can lead to bleeding disorders, so supplemental vitamin K is often given to prevent this.
Phytonadione Injectable Emulsion Prefilled Syringes contain a concentration of 1 mg of phytonadione per 0.5 mL of solution. When administered, this injectable emulsion works by increasing the levels of vitamin K in the blood, which in turn helps to produce important clotting factors.
The emulsion is prefilled in syringes for convenience and ease of administration in the neonatal population. This formulation allows for accurate dosing and reduces the risk of dosing error.
Overall, the Phytonadione Injectable Emulsion Prefilled Syringes help to maintain proper blood clotting in neonatal patients and prevent potential bleeding disorders.
- Primary indication: prophylaxis against Vitamin K Deficiency Bleeding (VKDB) in neonates - administered as a single 1 mg intramuscular dose at birth per AAP guidelines
- Secondary indication: treatment of confirmed vitamin K deficiency bleeding in neonatal patients under clinical supervision
- Intended for use in hospital delivery units, neonatal intensive care units (NICUs), birthing centres, and paediatric clinical settings
- Prefilled syringe format eliminates bedside preparation from multi-dose vials, reducing preparation time and dosing error risk in high-volume delivery settings
- Each syringe delivers the full standard prophylactic dose (1 mg) in a single 0.5 mL administration - no dilution or reconstitution required
- Clinicians transitioning from Aquamephyton (discontinued Merck formulation) will find the IMS phytonadione injectable emulsion at the same 2 mg/mL concentration directly compatible with existing neonatal protocols
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
- For use only by or under the supervision of a licensed healthcare professional - RX designation applies; not for general public administration
- Contains polysorbate 80 and propylene glycol as emulsifying excipients - review full inactive ingredient list in the manufacturer product insert prior to administration in neonates with known hypersensitivity to these components
- Severe hypersensitivity reactions, including anaphylaxis, have been reported with intravenous administration of phytonadione injectable emulsion - the intramuscular route is standard for neonatal prophylaxis; IV use requires clinical justification and monitoring
- Do not administer IV push rapidly - if IV administration is clinically necessary, refer to current prescribing information for recommended dilution and infusion rate
- Store per manufacturer specifications - refer to product insert for temperature requirements and light-sensitivity guidance; storage conditions not specified in provided data
- Discard unused portion - single-dose prefilled syringe format; do not retain for re-use
- All safety, contraindication, and adverse event information should be verified against the current FDA-approved prescribing information supplied with the product
This product is intended for use by qualified healthcare professionals or under the guidance of a licensed medical provider. It is not a substitute for professional medical advice, diagnosis, or treatment.
This product is a prescription (RX) pharmaceutical and is available for purchase only by licensed healthcare professionals, accredited medical facilities, and authorised institutional buyers in the United States. Mountainside Medical Equipment supplies this product exclusively through verified wholesale channels.
The clinical information provided on this page is intended for informational reference by qualified healthcare professionals only. It does not constitute medical advice and is not a substitute for the manufacturer's current FDA-approved prescribing information, package insert, or the independent clinical judgment of a licensed medical provider.
All product claims, ingredient information, storage requirements, and dosing guidance should be verified against the official IMS product insert supplied with this product prior to clinical use. Mountainside Medical makes no independent clinical efficacy or safety claims beyond those established in the manufacturer's prescribing information.
This product has not been evaluated by the FDA for any use beyond those described in the approved prescribing information. Do not use this product outside of its approved indications without appropriate clinical oversight.
Healthcare professionals should consult current AAP guidelines, CDC recommendations, and the FDA-approved prescribing information for complete clinical guidance on phytonadione use in neonatal patients.