Filters
- 0.9% benzyl alcohol (1)
- Aesthetic Supplies MedSpa Supplies & Cosmetic Surgery Supplies (7)
- Bac (1)
- bac water (1)
- Bacterio (1)
- bacteriostatic (2)
- bacteriostatic water (2)
- Diluent (3)
- dilution (1)
- doctor-only (34)
- Hospira Pfizer (8)
- Multiple dose Sterile water (1)
- Multiple Dose Vial (4)
- reconstitution (1)
- Sale (1)
- Static water (1)
- sterile water (1)
- Sterile Water For Injection (5)
- sterile water injection (1)
- water for diluting (2)
- water for injection (5)
- Electrolyte balance (1)
- Hospira Vials (8)
- Intravenous (4)
- Intravenous Solution (3)
- Medication delivery (1)
- nacl (1)
- Rehydration (1)
- Sodium Chloride (4)
- Sodium Chloride 0.9 (2)
- Subcutaneous (1)
- 1% lidocaine (2)
- Lidocaine (8)
- Lidocaine 1% (3)
- Lidocaine HCl (3)
- lidocaine hydochloride (6)
- Lidocaine Hydrochloride (7)
- Lidocaine Injection (4)
- Lidociane for injection (3)
- local anesthetic (3)
- numbing (2)
- Pfizer Lidocaine (2)
- 10ml Sterile Vials (1)
- Empty (3)
- Empty Glass Vial (2)
- Empty Glass Vials (2)
- Empty Vial (3)
- empty vials (1)
- Flip-Top Caps (1)
- glass vial (1)
- Injectable Solutions (1)
- Injection Vials (2)
- Laboratory (1)
- Laboratory Supplies (1)
- Pharmacy Compounding (2)
- Sterile water 20 ml (1)
- Sterile water Injection (2)
- blood disorders (1)
- Corticosteroid (1)
- Corticosteroid Medication (1)
- Corticosteroid Medicine (1)
- Corticosteroids (1)
- Depo (1)
- Depo Medrol (1)
- Depo-Medrol Injection (1)
- Glucocorticoid Corticosteroid (1)
- Joint Pain Treatment (1)
- Methylprednisolone (1)
- Methylprednisolone Acetate (1)
- Synthetic Corticosteroid (1)
- 5% Dextrose (1)
- Blood sugar (1)
- Carbohydrate source (1)
- Dextrose 50 (1)
- Dextrose For Injection (1)
- glucose (1)
- Hypertonic Solution (1)
- Hypoglycemia (1)
- Hypoglycemia Treatment (1)
- Intravenous Administration (1)
- Severe Hypoglycemia (1)
- bulk lidocaine (2)
- cosmetic anesthesia (1)
- dental anesthesia (2)
- dental injection (1)
- Epinephrine (4)
- Epinephrine Injection (4)
- hospital anesthesia (1)
- injectable anesthesia (1)
- Lidocaine 2% with epinephrine (1)
- Lidocaine with Epinephrine (2)
- Local Anesthetic (3)
- nerve block (3)
- Nerve blocker (4)
- Pain Management (1)
- vasoconstrictor injection (1)
- Bacteriostatic Saline (1)
- Bacteriostatic Sodium Chloride (1)
- Intramuscular (2)
- Intramuscular Injection (1)
- Normal Saline (1)
- Preservative (1)
- Sodium Chloride 0.9% (1)
- Single Dose (2)
- Sterile Water 100 ml (1)
- 50 ml (1)
- Single-use (1)
- Sterle Water for Injection (1)
- 2% lidocaine (2)
- hospital pharmacy (1)
- Lidocaine2 percent (2)
- numb pain (2)
- numbing shots (1)
- pain relief injection (1)
- surgical anesthesia (1)
- Alprostadil (1)
- alprostadil for injection (1)
- Alprostadil Kit (1)
- Caverject (1)
- Caverject Impulse Alprostadil Kit (1)
- ED treatment (1)
- erectile dysfunction (1)
- Erectile Dysfunction Treatment (1)
- Prostin VR (1)
- Treat Erectile Dysfunction (1)
- Treat Impotence (1)
- Antineoplastic Agent (1)
- Buy Carboplatin (1)
- Carboplatin 150 mg (1)
- Carboplatin for Injection (2)
- Carboplatin Near Me (1)
- Chemotherapeutic Medication (1)
- chemotherapy medications (2)
- Lung Cancer (2)
- Ovarian Cancer (1)
- Paraplatin (1)
- Prostate Cancer (1)
- treat ovarian cancer (2)
- Treat Prostate Cancer (1)
- 8.4% Sodium Bicarbonate (1)
- Extracorporeal Circulation of Blood (1)
- Metabolic Acidosis (1)
- Severe Dehydration (1)
- Sodium Bicab (1)
- sodium bicarbonate (1)
- Sodium Bicarbonate for Injection (1)
- Sodium Hydrogen Carbonate (1)
- Treatment of metabolic acidosis (1)
- 1259501 (1)
- biofilm formation (1)
- blood reflux occlusion (1)
- connector (1)
- iv connector (1)
- lifeshield (1)
- neutral pressure (1)
- neutron catheter (1)
- patency device (1)
- Bladder Cancer (1)
- BAGS (1)
- Intravenous infusion (1)
- IV bag (1)
- IV Solution (1)
- IV Therapy (1)
- NACL (1)
- normal saline (1)
- Saline Solution (1)
- 2049-02 (1)
- Carpuject (1)
- Carpuject Syringe System (1)
- Cartridge Holder (1)
- Connective Tissue (1)
- copper (1)
- Copper Deficiency (1)
- Copper for Injection (1)
- Copper Injection (1)
- Cupric Chloride (1)
- Iron Metabolism (1)
- Malabsorption (1)
- Menkes Disease (1)
- Short Bowel Syndrome (1)
- Total Parenteral Nutrition (1)
- Trace element (1)
- Treat Copper Deficiencies (1)
- Gel Flow Syringe (1)
- Gel-Flow NT (1)
- hemostatic (1)
- 30 ml (2)
- Empty Vials (2)
- Mixing Vial (2)
- nonpyrogenic (2)
- Empty Bags (1)
- Empty IV Bags (1)
- mixing medications (1)
- Blood Sugar Imbalances (1)
- Chromic Chloride (1)
- Chromium (1)
- Chromium for Injection (1)
- Fat Metabolism (1)
- insulin Function (1)
- Mineral Chromium (1)
- Analgesia (1)
- Intramuscularly (1)
- Ketorolac for Injection (1)
- Ketorolac Shot (1)
- Ketorolac Tromethamine (1)
- ketorolac tromethamine injection (1)
- Moderate to severe pain (1)
- Nonsteroidal anti-inflammatory drugs (NSAIDs) (1)
- Pain Relief (1)
- Prostaglandins (1)
- Short-term management (1)
- Surgical procedures (2)
- Toradol (1)
- Acute agitation treatment (1)
- Agitation (1)
- Agitation Treatment (1)
- antipsychotic (1)
- antipsychotic for ER (1)
- Antipsychotic medication (1)
- Bipolar Disorder (1)
- Emergency mental health (1)
- Geodon for Injection (1)
- Geodon injection (1)
- Geodon injection for agitation (1)
- Injectable antipsychotic (1)
- mental health treatment (1)
- Pfizer Hospira (1)
- psychiatric medication (1)
- Rapid-acting antipsychotic (1)
- Schizophrenia (1)
- Severe agitation control (1)
- Treat bipolar disorder (1)
- Treat schizophrenia (1)
- Viatris specialty injectables (1)
- Ziprasidone (1)
- Ziprasidone injection (1)
- anticoagulant (1)
- Atrial fibrillation (1)
- blood clots (1)
- Blood clotting (1)
- blood thinner injection (1)
- Break up Blood Clots (1)
- cardiac surgery (1)
- Deep Vein Thrombosis (1)
- dialysis anticoagulant (1)
- Heparin (1)
- Heparin 10000 unit (1)
- Heparin Sodium (1)
- Heparin-Induced Thrombocytopenia (1)
- hospital anticoagulant (1)
- intravenous Lines (1)
- IV heparin (1)
- Prevent Blood Clots (1)
- Prevention of clotting (1)
- pulmonary embolism (1)
- pulmonary embolism treatment (1)
- Sodium Heparin (1)
- subcutaneous heparin (1)
- treatment of venous thrombosis (1)
- 5 Dextrose (1)
- Dextrose 5% (1)
- Dextrose 5% and 0.9% Sodium Chloride (1)
- dextrose with sodium chloride (1)
- Iv Solution (1)
- Sugar Water (1)
- 10 ml (1)
- 10 ml Sterile water (1)
- Sterile Water 10 ml (1)
- Sterile Water Injection (1)
- Epinephrine Vials (2)
- EpiPen (1)
- ou-exclude (2)
- Bupivacaine (2)
- Bupivacaine 0.75% (1)
- Bupivacaine for Injection (2)
- Bupivacaine Hydrochloride (1)
- Bupivacaine Injection (1)
- Dental Procedures (1)
- Obstetric Procedures (1)
- Postoperative Recovery (1)
- Surgical Procedures (1)
- Bupivacaine Hydrochloride and Epinephrine (1)
- Marcaine (1)
- Anorexia (1)
- DNA synthesis (1)
- Dysosmia (1)
- Geophagia (1)
- Growth Retardation (1)
- Hepatosplenomegaly (1)
- Hypogeusia (1)
- Hypogonadism (1)
- Immune Function (1)
- Metabolic Function (1)
- Parakeratosis (1)
- Protein Synthesis (1)
- Trace Element supplement (1)
- Treatment of anorexia (1)
- Wound Healing (1)
- zinc (1)
- Zinc Chloride (1)
- Zinc deficiency (1)
- Zinc for Injection (1)
- Lidocaine 2% Injection 50 mL (1)
- Numbing (1)
- Compounding Kit (1)
- Laboratory samples (1)
- Laboratory settings (1)
- Medisca (1)
- Reconstitution (1)
- Antioxidant Enzyme (1)
- Bone formation (1)
- Enzymes (1)
- Manganese (1)
- Manganese Chloride (1)
- Manganese deficiency (1)
- Manganese Injection (1)
- metabolism (1)
- Toxicity (1)
- TPN (1)
- Trace Element Supplement (1)
- Treat Manganese Deficiencies (1)
- Furosemide (2)
- Lasix (2)
- Treat Edema (1)
- Treat Fluid Retention (2)
- Treat Hypertension (1)
- Treat Liver Disease (2)
- Anesthesia (1)
- Local Anesthesia (1)
- Xylocaine (1)
- edema (1)
- Treat edema (1)
- Treat Heart failure (1)
Hospira
Hospira
Hospira, Inc. was an American specialty pharmaceutical and medical-device company founded as a spin‐off of Abbott Laboratories’ hospital products division. Headquartered in Lake Forest, Illinois, Hospira specialized in hospital-focused products – notably generic injectable drugs and medication-delivery systems (such as infusion pumps, IV sets and needle-free syringes). Before its acquisition, Hospira was the world’s largest producer of generic sterile injectables, serving hospitals, clinics and long-term care facilities worldwide. It had on the order of 18–19,000 employees and annual revenues around $4–4.5 billion (circa 2014). Importantly, Hospira also developed biosimilar (generic biologic) products and was described as “a global leader in biosimilars”.
Key milestones in Hospira’s history:
- May 3, 2004: Hospira began trading on the NYSE as an independent company, following Abbott Laboratories’ spin-off of its hospital products business. (Abbott announced that the “previously announced separation” of its hospital products unit would create Hospira as a new company.)
- September 2015: Pfizer completed its acquisition of Hospira. In its press release, Pfizer highlighted that Hospira was “the world’s leading provider of injectable drugs and infusion technologies and a global leader in biosimilars”, and it integrated Hospira’s portfolio of generics and infusion products into Pfizer’s established pharma business.
- October 2016: Pfizer sold off Hospira’s infusion systems business (IV pumps, infusion sets, solutions, etc.) to ICU Medical for $1.0 billion. After this deal, ICU Medical became the owner of Hospira’s former infusion-device products, while Pfizer retained Hospira’s portfolio of generic sterile injectable drugs and biosimilars.
Products and Business
Before its acquisition, Hospira’s product lines centered on medications and devices used in hospitals. Its pharmaceutical division produced a broad range of generic sterile injectables – for example, products for anesthesia, oncology, critical care and pain management – as well as biosimilars (such as Retacrit™, a biosimilar epoetin). Concurrently, its medical-device division manufactured medication-delivery systems: this included electronic infusion pump systems, IV administration sets, and related monitoring and medication-management technology. These products were widely used in U.S. hospitals (Hospira earned ~85% of its revenues from U.S. sales), but also sold internationally.
By the mid-2010s, Hospira’s combined business made it a leader in sterile injectable drugs globally. A Pfizer executive noted at acquisition that Hospira’s products and expertise “add to” Pfizer’s portfolio of hospital drugs and biosimilars. In short, Hospira’s legacy was that of a major hospital-product manufacturer – its generics and infusion systems supplied millions of doses and infusions in healthcare settings worldwide.
Acquisition by Pfizer
On September 3, 2015, Pfizer announced that it had completed the takeover of Hospira. The acquisition was valued at over $15 billion and gave Pfizer control of Hospira’s generic-products and infusion-device businesses. In the official press release, Pfizer’s CEO stated: “Hospira, the world’s leading provider of injectable drugs and infusion technologies… is now part of Pfizer”. Pfizer planned to integrate Hospira into its Global Established Pharmaceuticals (GEP) business, strengthening its sterile-injectables portfolio and biosimilars lineup.
The deal folded Hospira’s results into Pfizer’s financial statements. After closing, Hospira ceased to operate as an independent company – its name lived on only within the larger Pfizer organization. Pfizer continued to market many of Hospira’s former products under its own banner; for example, Hospira’s generic sterile injectables became part of Pfizer’s hospital drug offerings.
Sale of Infusion Systems to ICU Medical
Shortly after the Pfizer acquisition, the infusion-pump side of Hospira changed hands again. In October 2016, Pfizer agreed to sell Hospira’s global Hospira Infusion Systems business – including IV pumps, infusion solutions, and related devices – to ICU Medical. The deal, valued at $1 billion in cash and stock, was completed to create a focused “pure-play infusion therapy company”. As a result, ICU Medical absorbed all of Hospira’s infusion-pump products and technology. (ICU Medical’s CEO noted that Hospira and ICU had been collaborating for years and that combining the infusion portfolios would benefit customers.)
After this divestiture, Hospira’s infusion-pump legacy continued under the ICU Medical brand. For example, ICU Medical continues to develop and sell IV pumps and systems originally designed by Hospira, though now rebranded and integrated into ICU’s portfolio. Meanwhile, Pfizer retained Hospira’s generic-injectables and biosimilars portfolio as part of its hospital/sterile injectables business.
Current Status
Hospira no longer exists as a standalone company. Its former businesses now reside in two different entities:
- Pfizer: The company’s generic sterile injectable drugs (e.g. anesthetics, oncology injectables, biosimilars) are now part of Pfizer’s hospital and generics divisions. (Pfizer absorbed Hospira’s manufacturing and R&D for these products.)
- ICU Medical: The infusion pump and IV solutions business (previously “Hospira Infusion Systems”) is part of ICU Medical. ICU continues to market advanced infusion pumps, IV therapy sets and related devices that originated from the Hospira product line.
In summary, Hospira grew from an Abbott spin-out in 2004 to a leading hospital-products company, but by 2016 it had been fully integrated into Pfizer and ICU Medical. Its legacy lives on through the hundreds of generic injectables and infusion systems that continue to be widely used in hospitals today.
-
 Sale 20%
Original price $ 379.00Current price $ 305.00
Sale 20%
Original price $ 379.00Current price $ 305.00Bacteriostatic Water Injection by Hospira, Multiple Dose Vials 30 mL 25/Pack (Rx)
1115 reviewsBacteriostatic Water for Injection is a pharmaceutical-grade sterile diluent formulated with 0.9% benzyl alcohol as a preservative, enabling safe m...
View full details🔒 Medical License Required -
 Sale 19%
Original price $ 129.00Current price $ 105.00
Sale 19%
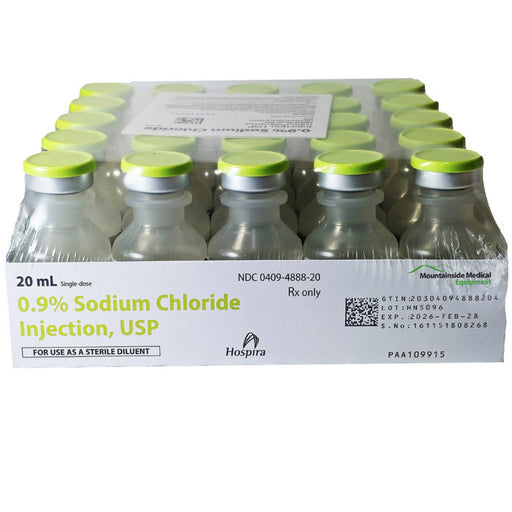
Original price $ 129.00Current price $ 105.00Sodium Chloride 0.9% For Injection 20 mL Vials 25/tray (Rx) Expiration : 10/31/26
40 reviewsSodium Chloride 0.9% For Injection 20 mL Vials are primarily used as a sterile isotonic solution for rehydration, electrolyte balance, and as a veh...
View full details🔒 Medical License Required -
 Sale 23%
Original price $ 179.99Current price $ 139.00
Sale 23%
Original price $ 179.99Current price $ 139.00Lidocaine 1%, Lidocaine Injection 20 mL Multiple Dose Vial Hospira 25/Pack (Rx)
23 reviewsLidocaine 1% Injection Lidocaine (also called lignocaine) 1% injection is a local anesthetic solution (10 mg of lidocaine per mL) used to numb tiss...
View full details🔒 Medical License Required -
 Sale 28%
Original price $ 39.95Current price $ 28.95
Sale 28%
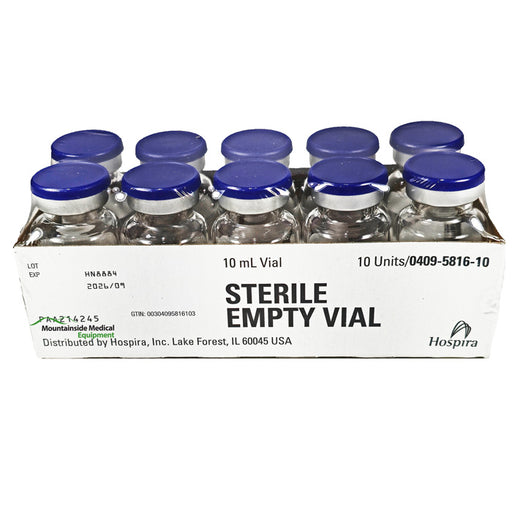
Original price $ 39.95Current price $ 28.95Sterile Empty Vials 10 mL Glass with Flip-Top Caps 10/Box
8 reviewsSterile Empty Vials 10 mL are primarily utilized in pharmaceutical and laboratory settings for the safe storage, handling, and transport of injecta...
View full details -
 Sale 20%
Original price $ 275.00Current price $ 219.00
Sale 20%
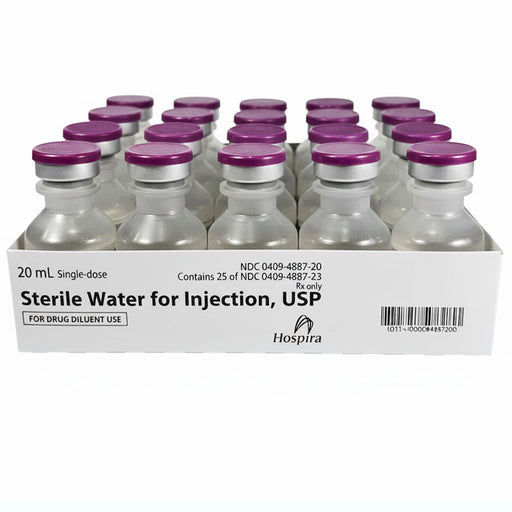
Original price $ 275.00Current price $ 219.00Sterile Water Injection 20 mL Single-Dose Vials by Hospira 25/Tray (Rx)
54 reviewsSterile Water for Injection 20 mL by Pfizer is a pharmaceutical-grade solvent used to dilute or dissolve medications for injection. It is employed ...
View full details🔒 Medical License Required -
 Sale 41%
Original price $ 99.95Current price $ 59.00
Sale 41%
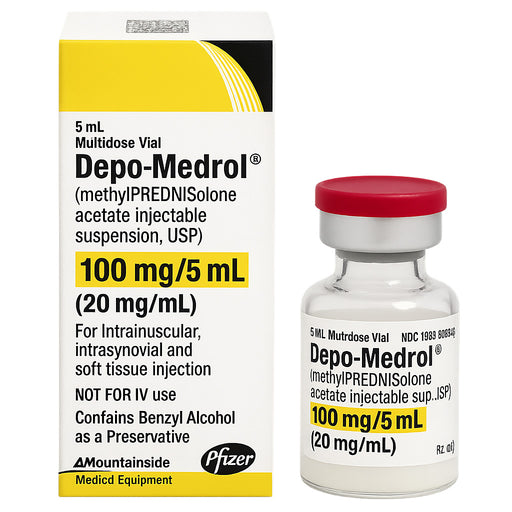
Original price $ 99.95Current price $ 59.00Depo-Medrol Injection Methylprednisolone Acetate 100 mg/5 mL (20 mg/mL) Multiple Dose Vials 5 mL (Rx)
No reviewsPfizer Injectables | Fast Relief from Inflammation & Autoimmune Disorders Depo-Medrol Injection Methylprednisolone 100 mg/5 mL (20 mg/mL) Multi...
View full details🔒 Medical License Required -
 Sale 13%
Original price $ 297.00Current price $ 259.00
Sale 13%
Original price $ 297.00Current price $ 259.00Dextrose 50% Injection Vials 50 mL by Pfizer 25/tray (Rx)
5 reviewsSterile Water for Injection 100 mL Glass Vials are used as a solvent or diluent for the preparation of medications intended for injection. This ste...
View full details🔒 Medical License Required -
 Sale 16%
Original price $ 525.00Current price $ 443.00
Sale 16%
Original price $ 525.00Current price $ 443.00Lidocaine 2% with Epinephrine 2% 1:100,000 Injection 50 mL Multiple Dose, 25/Pack (Rx) Expiration 1/31/27
No reviewsLidocaine 2% with Epinephrine 1:100,000 for Injection is a combination local anesthetic and vasoconstrictor solution commonly used in medical and d...
View full details🔒 Medical License Required -
 Sold out
Original price $ 119.00Current price $ 95.00
Sold out
Original price $ 119.00Current price $ 95.00Bacteriostatic Sodium Chloride 0.9% Injection Normal Saline Multiple Dose 20 mL, 25/pk (Rx)
92 reviewsBacteriostatic Sodium Chloride 0.9% Injection Multiple Dose Vials are used as a diluent for medications, providing a sterile solution for injection...
View full details -
 Sale 15%
Original price $ 445.00Current price $ 379.00
Sale 15%
Original price $ 445.00Current price $ 379.00Sterile Water for Injection 100 mL Glass Vials, 25/Tray (Rx)
1 reviewSterile Water for Injection 100 mL Glass Vials are used as a solvent or diluent for the preparation of medications intended for injection. This ste...
View full details🔒 Medical License Required -
 Sale 15%
Original price $ 385.00Current price $ 329.00
Sale 15%
Original price $ 385.00Current price $ 329.00Sterile Water for Injection 50 mL Single-Use Vials, 25/Tray (Rx)
30 reviewsSterile Water for Injection 50 mL Single-Use Vials are used as a solvent or diluent for the preparation of medications for injection. These vials p...
View full details🔒 Medical License Required -
 Sale 40%
Original price $ 175.00Current price $ 105.00
Sale 40%
Original price $ 175.00Current price $ 105.00Lidocaine 2%, Lidocaine Injection Numbing Shot 20 mL Multi-Dose Vials Hospira Pfizer 25/tray (Rx)
7 reviewsLidocaine 2% Injection - Multi-Dose Lidocaine HCl 2% injection is an amide-type local anesthetic. The “2%” means 20 mg of lidocaine per mL of solut...
View full details🔒 Medical License Required -
 Sale 32%
Original price $ 1,395.95Current price $ 945.00
Sale 32%
Original price $ 1,395.95Current price $ 945.00Caverject Impulse (Alprostadil for Injection) 20 mcg, 6/Box (RX)
No reviewsCaverject Impulse is the brand name for a medication called Alprostadil for Injection. It contains the active ingredient alprostadil, which is a sy...
View full details🔒 Medical License Required -
 Sale 31%
Original price $ 85.95Current price $ 59.35
Sale 31%
Original price $ 85.95Current price $ 59.35Carboplatin Injection 150 mg / 15 mL Multiple Dose Vial 15 mL (RX)
No reviewsCarboplatin for Injection 150mg / 15 mL Multiple Dose Vial 15 mL is a form of chemotherapy medication used to treat various types of cancer, includ...
View full details🔒 Medical License Required -
 Sale 21%
Original price $ 495.00Current price $ 389.00
Sale 21%
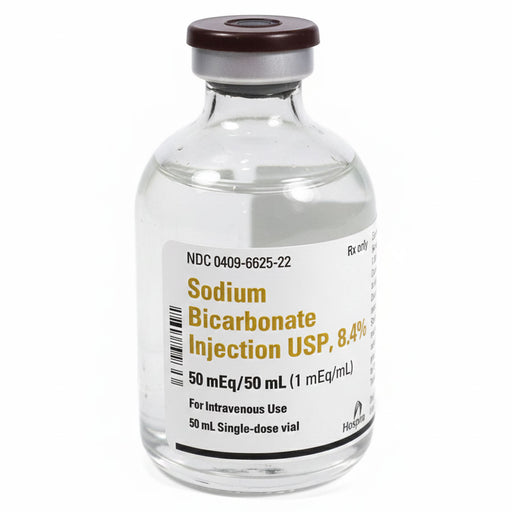
Original price $ 495.00Current price $ 389.00Sodium Bicarbonate Injection 8.4% Hospira 50 mL Vials 25/pack (Rx) Exp 3/31/27
4 reviewsSodium Bicarbonate Injection 8.4% Sodium bicarbonate 8.4% is a sterile, preservative-free hypertonic IV solution of sodium bicarbonate (NaHCO₃) at ...
View full details🔒 Medical License Required -
 Sale 10%
Original price $ 859.00Current price $ 769.00
Sale 10%
Original price $ 859.00Current price $ 769.00LifeShield Neutron Catheter Patency Device 100/Case
No reviewsBest IV Access Protection, Catheter Occlusion Prevention, and Infusion Therapy Safety Optimize patient safety and protect every intravenous line wi...
View full details -
 Sale 22%
Original price $ 159.95Current price $ 125.00
Sale 22%
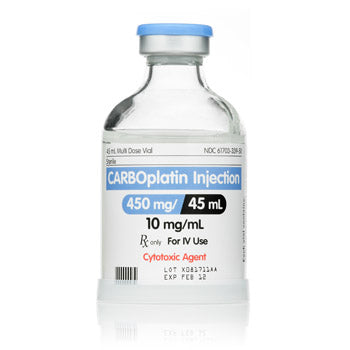
Original price $ 159.95Current price $ 125.00Carboplatin for Injection 450mg / 45 mL Multidose Vial 45mL, Preservative Free (RX)
No reviewsCarboplatin for Injection 450mg / 45 mL Multidose Vial 45 mL is a chemotherapy medication used to treat different types of cancer, including ovaria...
View full details🔒 Medical License Required -
 Sale 10%
Original price $ 14.95Current price $ 13.50
Sale 10%
Original price $ 14.95Current price $ 13.50IV Bag Sodium Chloride 0.9% IV Solution Saline 1000 mL Bag (EACH) (Rx)
173 reviewsSodium Chloride for Injection is a iv bags used as a iv solution. It is also made with clearly labeled graduation marks and has a rigid saddle with...
View full details🔒 Medical License Required -
 Sale 49%
Original price $ 0.49Current price $ 0.25
Sale 49%
Original price $ 0.49Current price $ 0.25Carpuject Syringe System, Cartridge Holder
1 reviewCarpuject Syringe Drug Delivery System features a reusable and autoclavable holder that is designed to hold prefilled syringes, making it quicker, ...
View full details -
 Sale 29%
Original price $ 2,650.00Current price $ 1,890.00
Sale 29%
Original price $ 2,650.00Current price $ 1,890.00Copper Injection (Cupric Chloride) Injection 10 mL, 25/Tray by Pfizer (Rx)
1 reviewCopper (Cupric Chloride) for Injection is used to treat or prevent copper deficiency in patients who cannot get enough copper through diet alone, o...
View full details🔒 Medical License Required -
 Sold out
Original price $ 345.95Current price $ 289.95
Sold out
Original price $ 345.95Current price $ 289.95Gel-Flow NT Absorbable Gelatin Powder Syringe Hemostatic Matrix for Flowable Applications (6 mL)
1 reviewThe Pfizer Gel-Flow NT device consists of 550mg of Gel-Foam® Sterile Powder (absorbable gelatin powder from absorbable gelatin sponge, USP), a syr...
View full details -
 Sale 14%
Original price $ 76.95Current price $ 65.95
Sale 14%
Original price $ 76.95Current price $ 65.955 mL Empty Glass Medication Vials, Sterile, Clear 25 Count
2 reviews5 mL Empty Glass Medication Vials, Sterile, are used for storing and preserving injectable medications, laboratory samples, or compounding ingredie...
View full details -
 Sold out
Original price $ 499.99Current price $ 369.00
Sold out
Original price $ 499.99Current price $ 369.00Empty IV Bag Container 1000 mL, Hospira
2 reviewsHospira Empty IV Bag Container is a single-use I.V. container for admixing compatible medications. *Rolling Manufacture Backorder* Use aseptic tec...
View full details -
 Sale 25%
Original price $ 1,450.00Current price $ 1,085.00
Sale 25%
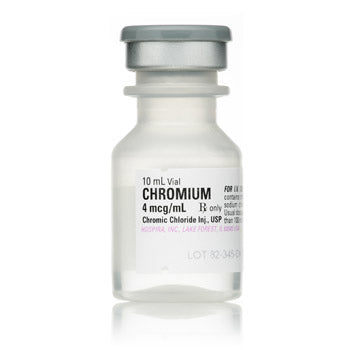
Original price $ 1,450.00Current price $ 1,085.00Chromium (Chromic Chloride) for Injection 10ml Vials, 25/Tray (RX)
No reviewsChromium (Chromic Chloride) for Injection is a medication used to supplement and maintain adequate levels of the mineral chromium in the body. It i...
View full details🔒 Medical License Required -
 Sold out
Original price $ 185.00Current price $ 39.95
Sold out
Original price $ 185.00Current price $ 39.95Ketorolac Tromethamine Injection 60 mg/2 mL Single Dose Vials 2 mL, 25/Tray (Rx)
1 reviewKetorolac Tromethamine Injection 30 mg/2 mL is used for the short-term management of moderate to severe pain, often following surgical procedures. ...
View full details -
 Sale 31%
Original price $ 1,295.95Current price $ 895.00
Sale 31%
Original price $ 1,295.95Current price $ 895.00Geodon for Injection (Ziprasidone) 20mg, 10/box (Rx)
No reviewsRapid-Acting Antipsychotic for Acute Agitation and Schizophrenia Geodon for Injection (Ziprasidone) 20mg by Viatris Specialty offers powerful, rapi...
View full details🔒 Medical License Required -
 Sale 27%
Original price $ 259.00Current price $ 189.00
Sale 27%
Original price $ 259.00Current price $ 189.00Heparin Sodium Injection 10,000 USP Multiple-Dose Vials 10 mL x 25/Pack (RX)
2 reviewsHeparin Sodium Injection 10,000 Heparin sodium injection is a prescription anticoagulant (blood thinner) given by subcutaneous (SC) or intravenous ...
View full details🔒 Medical License Required -
 Sold out
Original price $ 18.90Current price $ 12.40
Sold out
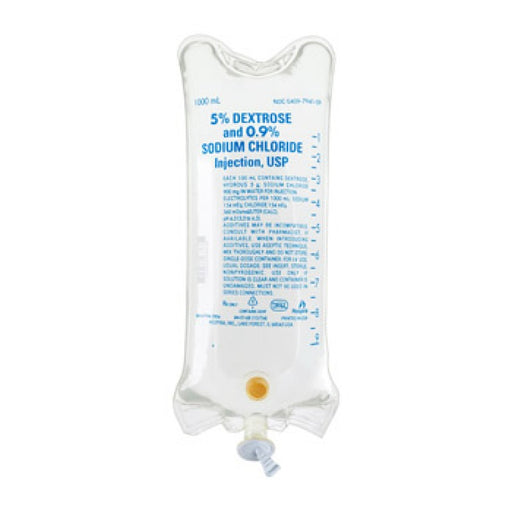
Original price $ 18.90Current price $ 12.40IV Bag Dextrose 5% and 0.9% Sodium Chloride IV Solution, 1000 mL Bag (Rx)
31 reviewsICU Medical 5% Dextrose and 0.9% Sodium Chloride injection, USP solutions are sterile and nonpyrogenic. They are large volume parenteral solutions ...
View full details -
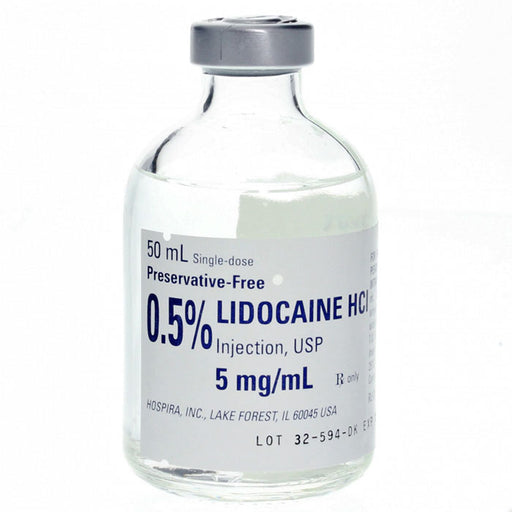 Sale 22%
Original price $ 249.95Current price $ 195.00
Sale 22%
Original price $ 249.95Current price $ 195.00Lidocaine Hydrochloride 0.5% Injection 50 mL Single-Dose, Preservative-Free 25/tray (Rx)
4 reviewsLidocaine Hydrochloride 0.5% for Injection 50 mL is used as a local anesthetic to numb specific areas during minor surgical procedures or medical i...
View full details🔒 Medical License Required -
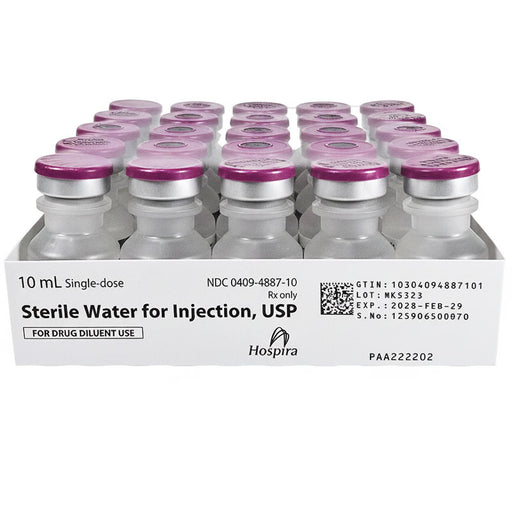 Sale 25%
Original price $ 219.95Current price $ 165.00
Sale 25%
Original price $ 219.95Current price $ 165.00Sterile Water for Injection 10 ml Single-Dose Vials 25/Tray by Hospira (Rx)
3 reviewsHospira Sterile Water for Injection is a pharmaceutical-grade, non-pyrogenic sterile water intended for use as a solvent or diluent for the prepara...
View full details🔒 Medical License Required -
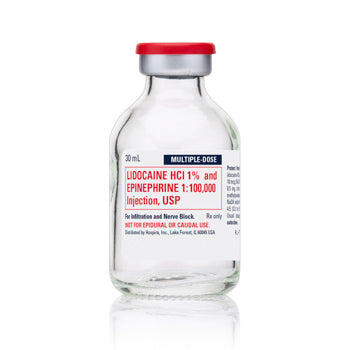 Sold out
Original price $ 598.00Current price $ 565.00
Sold out
Original price $ 598.00Current price $ 565.00Lidocaine 1% with Epinephrine 1% 1:100,000 Inj 30 mL Multiple Dose, 25/Pack (Rx) Exp :
No reviewsLidocaine 1% with Epinephrine 1:100,000 for Injection is used as a local anesthetic to provide pain relief and reduce bleeding during surgical, med...
View full details -
 Sale 14%
Original price $ 325.00Current price $ 279.00
Sale 14%
Original price $ 325.00Current price $ 279.00Lidocaine HCL 2% and Epinephrine 1% 1:100,000 for Injection 30 mL Multiple Dose, 25/Pack (Rx) Exp : 12/31/26
2 reviewsLidocaine HCL 2% and Epinephrine 1% 1:100,000 for Injection is used as a local anesthetic to provide pain relief during surgical or dental procedur...
View full details🔒 Medical License Required -
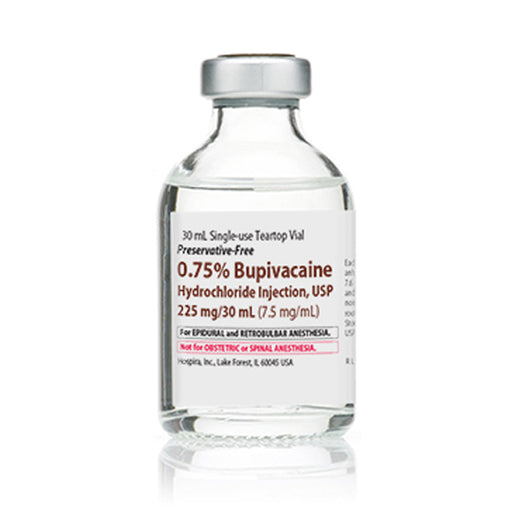 Sale 14%
Original price $ 229.95Current price $ 197.00
Sale 14%
Original price $ 229.95Current price $ 197.00Bupivacaine 0.75% for Injection Single-Dose Vials 30 mL, 25/Tray (Rx)
No reviewsBupivacaine 0.75% for Injection is a local anesthetic used to provide prolonged pain relief by blocking nerve signals during surgical, dental, or o...
View full details🔒 Medical License Required -
 Sale 28%
Original price $ 54.00Current price $ 38.95
Sale 28%
Original price $ 54.00Current price $ 38.95Marcaine (Bupivacaine Hydrochloride) 125 mg/50 mL with Epinephrine 1:200,000 Injection 0.25% Multi-Dose 50mL Vial (Rx)
1 reviewMarcaine (Bupivacaine Hydrochloride) 125 mg/50 mL with Epinephrine 1:200,000 is used for local anesthesia and pain management during surgical, dent...
View full details🔒 Medical License Required -
 Sale 15%
Original price $ 1,750.00Current price $ 1,495.00
Sale 15%
Original price $ 1,750.00Current price $ 1,495.00Zinc Chloride for Injection 1 mg/mL Single Dose 10 mL, 25/Tray
No reviewsZinc Chloride Injection is used as a trace element supplement in total parenteral nutrition (TPN) to prevent or treat zinc deficiency. It is essent...
View full details🔒 Medical License Required -
 Sale 17%
Original price $ 199.95Current price $ 165.00
Sale 17%
Original price $ 199.95Current price $ 165.00Lidocaine 2% Injection 50 mL Multiple-Dose Vials Hospira 25/Pack (Rx) Exp 12/31/26
3 reviewsLidocaine 2% Injection Lidocaine 2% for Injection is a fast-acting, amide-type local anesthetic widely used for injectable anesthesia in minor surg...
View full details🔒 Medical License Required -
 Sale 25%
Original price $ 105.95Current price $ 79.00
Sale 25%
Original price $ 105.95Current price $ 79.0030 mL Empty Sterile Vials, Clear Glass, 25/Tray by Fresenius Kabi
No reviews30 mL Empty Sterile Vials Clear Glass are used for storing and handling medications, laboratory samples, and other sterile liquids. Made from clear...
View full details -
 Sale 20%
Original price $ 2,250.00Current price $ 1,795.00
Sale 20%
Original price $ 2,250.00Current price $ 1,795.00Manganese Chloride Injection IV Trace Element Supplementation 0.1 mg/mL SDV 10 mL x 25/Tray (RX)
No reviewsEssential IV Trace Element Supplement for Parenteral Nutrition, Deficiency Correction, and Total Parenteral Nutrition Protocols Manganese Chloride ...
View full details🔒 Medical License Required -
 Sale 27%
Original price $ 44.95Current price $ 32.98
Sale 27%
Original price $ 44.95Current price $ 32.98Furosemide for Injection 10mg/mL Single-Dose Vial 2mL, 25/tray (Rx)
No reviewsFurosemide for Injection 10mg/mL Single-Dose Vial 2mL, 25/Tray by Eugia US is a prescription medication used in hospitals and other healthcare sett...
View full details🔒 Medical License Required -
 Sale 48%
Original price $ 189.00Current price $ 99.00
Sale 48%
Original price $ 189.00Current price $ 99.00Lidocaine Hydrochloride 1% Injection 50 mL Multiple Dose, 25/Pack (Rx)
5 reviewsLidocaine Hydrochloride 1% for Injection is a local anesthetic agent used to provide temporary relief of pain associated with various conditions. I...
View full details🔒 Medical License Required -
 Sale 29%
Original price $ 54.95Current price $ 39.00
Sale 29%
Original price $ 54.95Current price $ 39.00Furosemide Injection 10 mg Single-dose Vial 2 mL, 25/Tray by Pfizer (RX)
No reviewsFurosemide for Injection is a highly effective medication used for various conditions that cause fluid retention in the body. This powerful diureti...
View full details🔒 Medical License Required
People Also Searched For
Hospira
Hospira, Inc. was an American specialty pharmaceutical and medical-device company founded as a spin‐off of Abbott Laboratories’ hospital products division. Headquartered in Lake Forest, Illinois, Hospira specialized in hospital-focused products – notably generic injectable drugs and medication-delivery systems (such as infusion pumps, IV sets and needle-free syringes). Before its acquisition, Hospira was the world’s largest producer of generic sterile injectables, serving hospitals, clinics and long-term care facilities worldwide. It had on the order of 18–19,000 employees and annual revenues around $4–4.5 billion (circa 2014). Importantly, Hospira also developed biosimilar (generic biologic) products and was described as “a global leader in biosimilars”.
Key milestones in Hospira’s history:
- May 3, 2004: Hospira began trading on the NYSE as an independent company, following Abbott Laboratories’ spin-off of its hospital products business. (Abbott announced that the “previously announced separation” of its hospital products unit would create Hospira as a new company.)
- September 2015: Pfizer completed its acquisition of Hospira. In its press release, Pfizer highlighted that Hospira was “the world’s leading provider of injectable drugs and infusion technologies and a global leader in biosimilars”, and it integrated Hospira’s portfolio of generics and infusion products into Pfizer’s established pharma business.
- October 2016: Pfizer sold off Hospira’s infusion systems business (IV pumps, infusion sets, solutions, etc.) to ICU Medical for $1.0 billion. After this deal, ICU Medical became the owner of Hospira’s former infusion-device products, while Pfizer retained Hospira’s portfolio of generic sterile injectable drugs and biosimilars.
Products and Business
Before its acquisition, Hospira’s product lines centered on medications and devices used in hospitals. Its pharmaceutical division produced a broad range of generic sterile injectables – for example, products for anesthesia, oncology, critical care and pain management – as well as biosimilars (such as Retacrit™, a biosimilar epoetin). Concurrently, its medical-device division manufactured medication-delivery systems: this included electronic infusion pump systems, IV administration sets, and related monitoring and medication-management technology. These products were widely used in U.S. hospitals (Hospira earned ~85% of its revenues from U.S. sales), but also sold internationally.
By the mid-2010s, Hospira’s combined business made it a leader in sterile injectable drugs globally. A Pfizer executive noted at acquisition that Hospira’s products and expertise “add to” Pfizer’s portfolio of hospital drugs and biosimilars. In short, Hospira’s legacy was that of a major hospital-product manufacturer – its generics and infusion systems supplied millions of doses and infusions in healthcare settings worldwide.
Acquisition by Pfizer
On September 3, 2015, Pfizer announced that it had completed the takeover of Hospira. The acquisition was valued at over $15 billion and gave Pfizer control of Hospira’s generic-products and infusion-device businesses. In the official press release, Pfizer’s CEO stated: “Hospira, the world’s leading provider of injectable drugs and infusion technologies… is now part of Pfizer”. Pfizer planned to integrate Hospira into its Global Established Pharmaceuticals (GEP) business, strengthening its sterile-injectables portfolio and biosimilars lineup.
The deal folded Hospira’s results into Pfizer’s financial statements. After closing, Hospira ceased to operate as an independent company – its name lived on only within the larger Pfizer organization. Pfizer continued to market many of Hospira’s former products under its own banner; for example, Hospira’s generic sterile injectables became part of Pfizer’s hospital drug offerings.
Sale of Infusion Systems to ICU Medical
Shortly after the Pfizer acquisition, the infusion-pump side of Hospira changed hands again. In October 2016, Pfizer agreed to sell Hospira’s global Hospira Infusion Systems business – including IV pumps, infusion solutions, and related devices – to ICU Medical. The deal, valued at $1 billion in cash and stock, was completed to create a focused “pure-play infusion therapy company”. As a result, ICU Medical absorbed all of Hospira’s infusion-pump products and technology. (ICU Medical’s CEO noted that Hospira and ICU had been collaborating for years and that combining the infusion portfolios would benefit customers.)
After this divestiture, Hospira’s infusion-pump legacy continued under the ICU Medical brand. For example, ICU Medical continues to develop and sell IV pumps and systems originally designed by Hospira, though now rebranded and integrated into ICU’s portfolio. Meanwhile, Pfizer retained Hospira’s generic-injectables and biosimilars portfolio as part of its hospital/sterile injectables business.
Current Status
Hospira no longer exists as a standalone company. Its former businesses now reside in two different entities:
- Pfizer: The company’s generic sterile injectable drugs (e.g. anesthetics, oncology injectables, biosimilars) are now part of Pfizer’s hospital and generics divisions. (Pfizer absorbed Hospira’s manufacturing and R&D for these products.)
- ICU Medical: The infusion pump and IV solutions business (previously “Hospira Infusion Systems”) is part of ICU Medical. ICU continues to market advanced infusion pumps, IV therapy sets and related devices that originated from the Hospira product line.
In summary, Hospira grew from an Abbott spin-out in 2004 to a leading hospital-products company, but by 2016 it had been fully integrated into Pfizer and ICU Medical. Its legacy lives on through the hundreds of generic injectables and infusion systems that continue to be widely used in hospitals today.









































